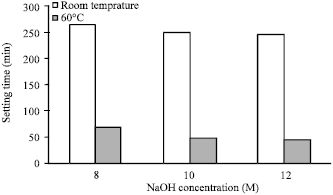
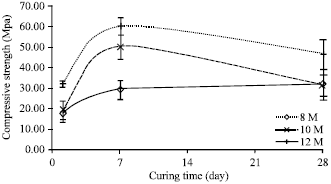
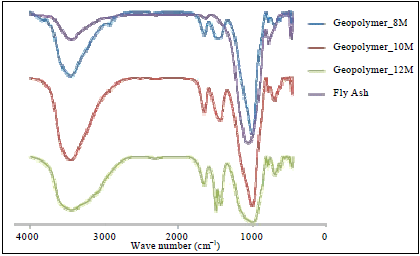
Research Article
Effect of Sodium Hydroxide Concentration on the Mechanical Property of Non Sodium Silicate Fly Ash Based Geopolymer
Department of Chemical Engineering, Tronoh, 31750, Perak, Malaysia
K.A. Azizli
Department of Chemical Engineering, Tronoh, 31750, Perak, Malaysia
Z. Man
Department of Chemical Engineering, Tronoh, 31750, Perak, Malaysia
T. Rahmiati
Department of Chemical Engineering, Tronoh, 31750, Perak, Malaysia
M.F. Nuruddin
Department of Civil Engineering, Universiti Teknologi Petronas, Bandar Sri Iskandar, Tronoh, 31750, Perak, Malaysia