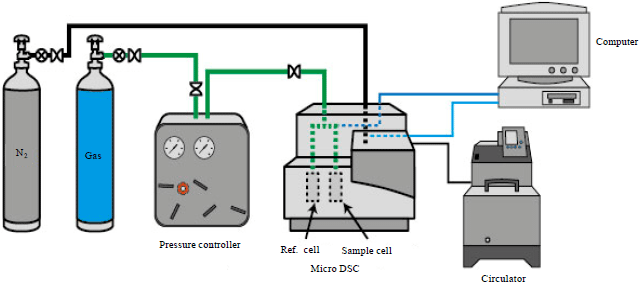
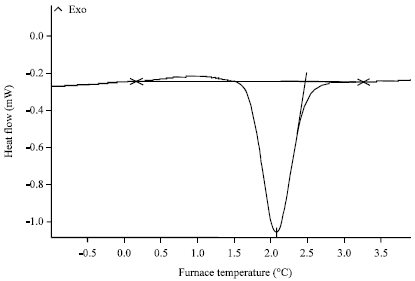
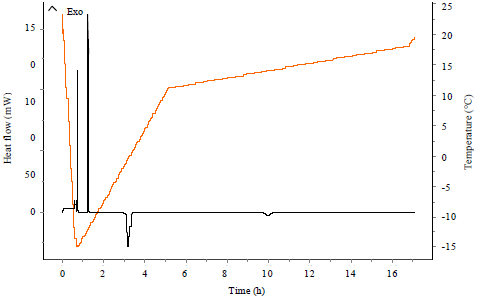
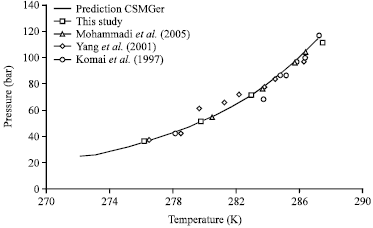
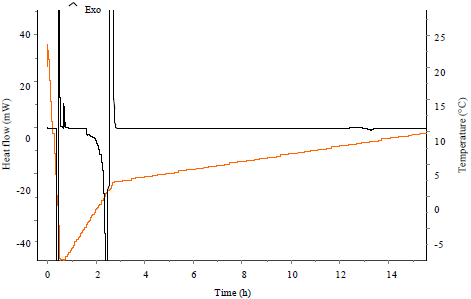
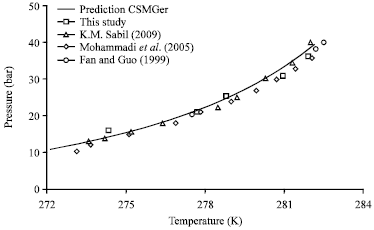
Research Article
Hydrate Equilibrium Measurement of Single CO2 and CH4 Hydrates Using Micro DSC
Department of Chemical Engineering, Universiti Teknology PETRONAS, Bandar Seri Iskandar, Tronoh, 31750, Perak, Malaysia
K.M. Sabil
Institute of Petroleum Engineering, Heriot-Watt University Malaysia, Putrajaya, 62100, Malaysia
L. Ismail
Department of Chemical Engineering, Heriot-Watt University Malaysia, Putrajaya, 62100, Malaysia
A. Jaafar
Department of Mechanical Engineering, Universiti Teknology PETRONAS, Bandar Seri Iskandar, Tronoh, 31750, Perak, Malaysia