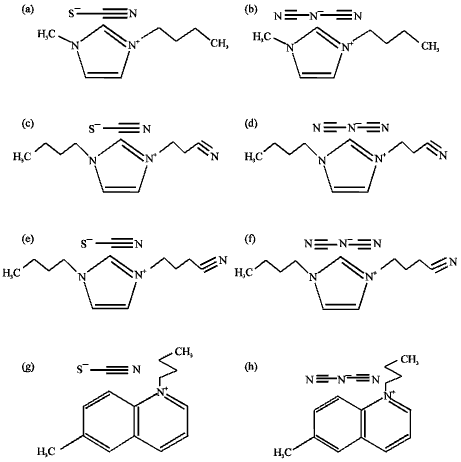
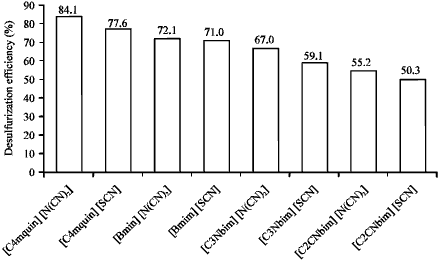
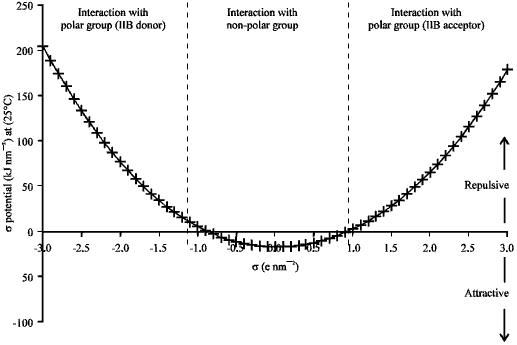
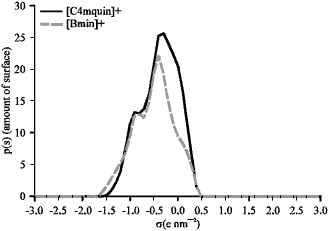
Research Article
Extraction of Dibenzothiophene from N-Dodecane using Ionic Liquids
Department of Chemical Engineering, Tronoh, Perak, Malaysia
M. Salleh
Department of Chemical Engineering, Tronoh, Perak, Malaysia
M. Ibrahim
Department of Chemical Engineering, Tronoh, Perak, Malaysia
Abd Mutalib
Department of Chemical Engineering, Tronoh, Perak, Malaysia
Cecilia Devi Wilfred
Department of Fundamental and Applied Science, Universiti Teknologi PETRONAS, Tronoh, Perak, Malaysia