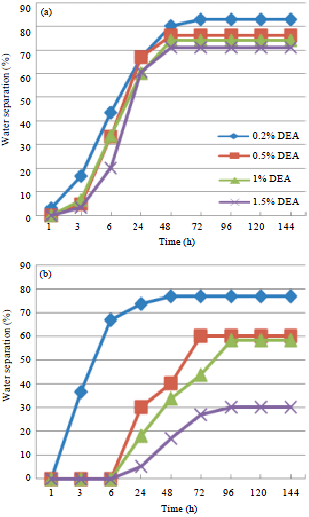
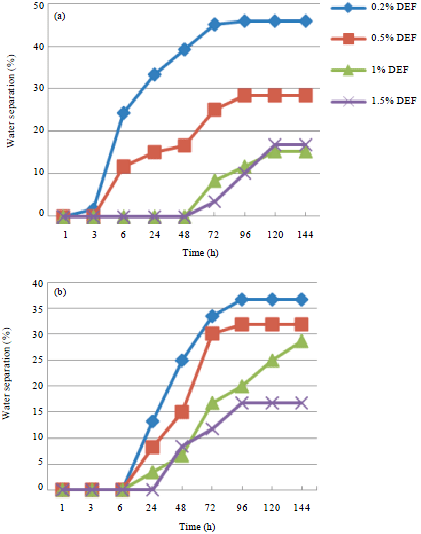
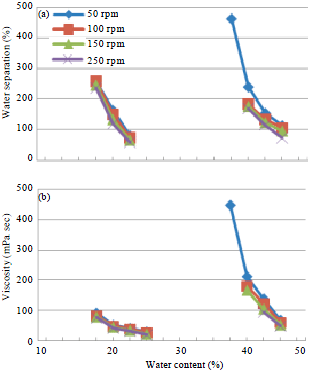
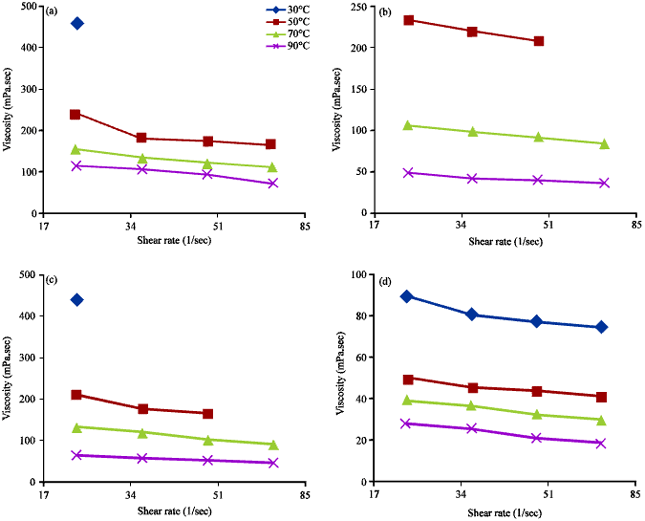
Research Article
Stability of Water-in-Crude Oil Emulsion Using Cocamide Surfactant
Chemical Engineering Programme, Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Lebuhraya Tun Razak, Gambang, 26300, Kuantan, Pahang, Malaysia
Abdurhman H. Nour
Chemical Engineering Programme, Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Lebuhraya Tun Razak, Gambang, 26300, Kuantan, Pahang, Malaysia
Ahmad Ziad Sulaiman
Chemical Engineering Programme, Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Lebuhraya Tun Razak, Gambang, 26300, Kuantan, Pahang, Malaysia