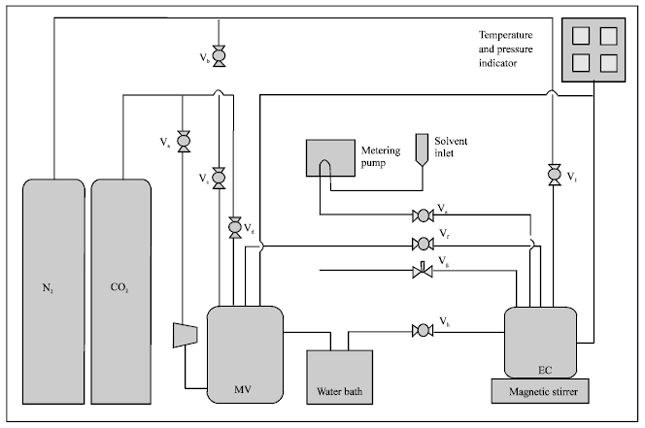
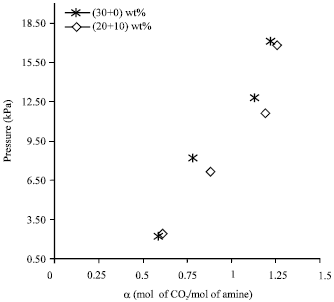
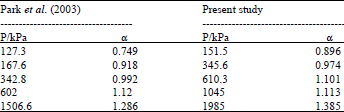
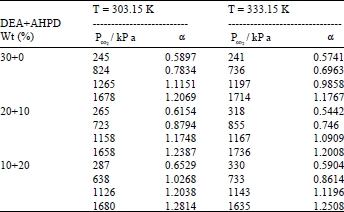
Research Article
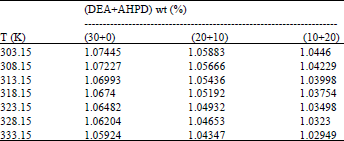
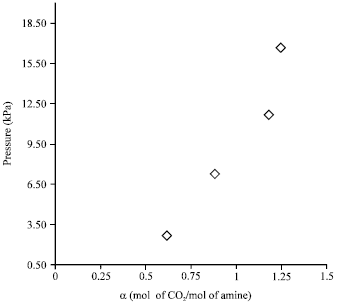
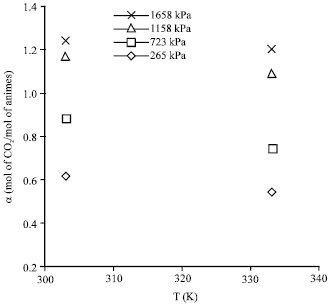
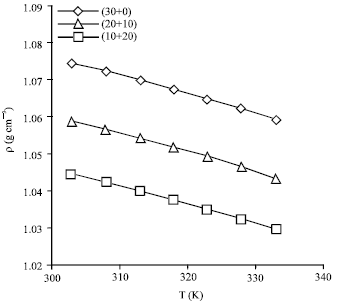
Absorption of Green House Gas (CO2) and Physical Properties of Aqueous Solutions of 2-Amino-2-Hydroxymethyl-1, 3-Propanediol and Di-Ethanolamine (AHPD+DEA) at Elevated Pressures
Research Centre for Carbon Dioxide Capture (RCCO2CTM), Universiti Teknologi PETRONAS, Tronoh, 31750, Perak, Malaysia
Azmi Mohd Shariff
Research Centre for Carbon Dioxide Capture (RCCO2CTM), Universiti Teknologi PETRONAS, Tronoh, 31750, Perak, Malaysia
Muhammad Azmi Bustam
Research Centre for Carbon Dioxide Capture (RCCO2CTM), Universiti Teknologi PETRONAS, Tronoh, 31750, Perak, Malaysia
Sami Ullah
Research Centre for Carbon Dioxide Capture (RCCO2CTM), Universiti Teknologi PETRONAS, Tronoh, 31750, Perak, Malaysia