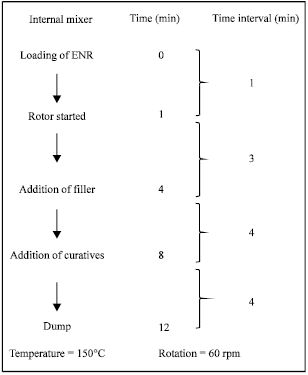
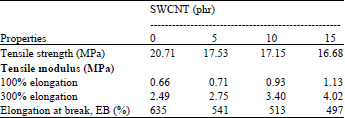
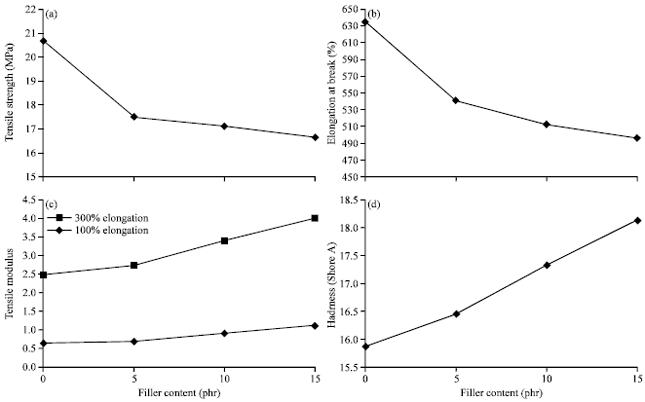
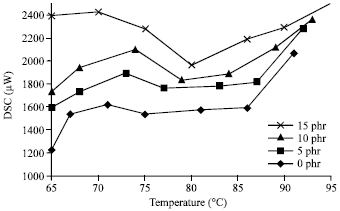
Research Article
Mechanical and Thermal Properties of Single-Walled Carbon Nanotube Filled Epoxidized Natural Rubber Nanocomposite
Faculty of Manufacturing Engineering, Universiti Teknikal Malaysia Melaka (UteM), Hang Tuah Jaya, Durian Tunggal, 76100, Melaka, Malaysia
E. Talib
Faculty of Manufacturing Engineering, Universiti Teknikal Malaysia Melaka (UteM), Hang Tuah Jaya, Durian Tunggal, 76100, Melaka, Malaysia
N. Mohamad
Faculty of Manufacturing Engineering, Universiti Teknikal Malaysia Melaka (UteM), Hang Tuah Jaya, Durian Tunggal, 76100, Melaka, Malaysia
M. S. Kasim
Faculty of Manufacturing Engineering, Universiti Teknikal Malaysia Melaka (UteM), Hang Tuah Jaya, Durian Tunggal, 76100, Melaka, Malaysia
M. W.A. Rashid
Faculty of Manufacturing Engineering, Universiti Teknikal Malaysia Melaka (UteM), Hang Tuah Jaya, Durian Tunggal, 76100, Melaka, Malaysia