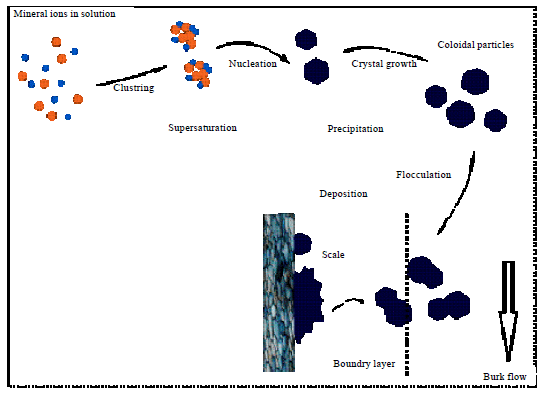
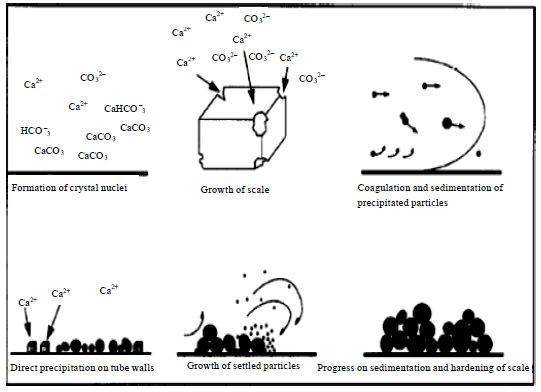
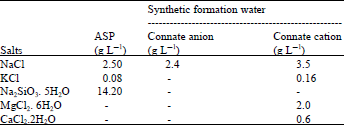
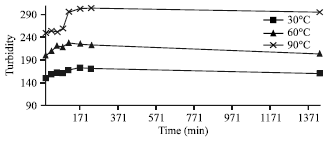
Research Article
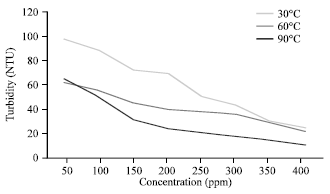
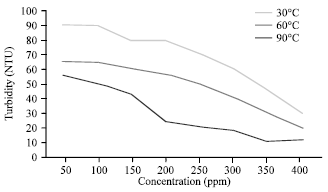
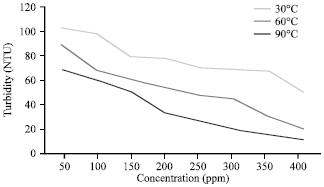
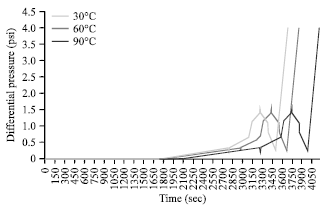
Effects of Temperature on Silicate Scale Inhibition During ASP Flooding
Department of Petroleum Engineering, Faculty of Petroleum Geosciences and Engineering, Universiti Teknologi PETRONAS, Seri Iskandar, Tronoh, 31750, Perak, Darul-Ridzuan, Malaysia
I.M. Saaid
Department of Petroleum Engineering, Faculty of Petroleum Geosciences and Engineering, Universiti Teknologi PETRONAS, Seri Iskandar, Tronoh, 31750, Perak, Darul-Ridzuan, Malaysia