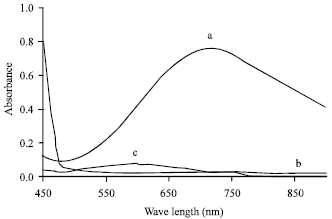
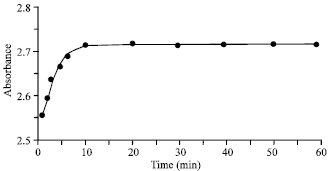
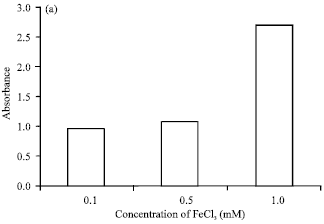
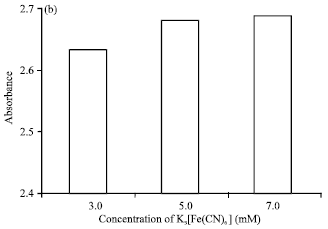
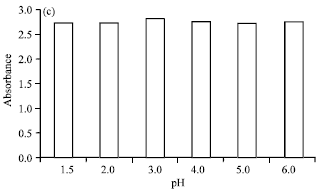
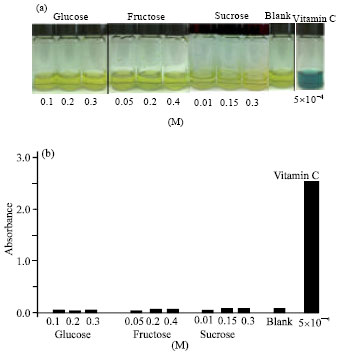
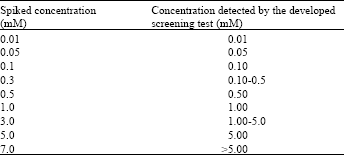
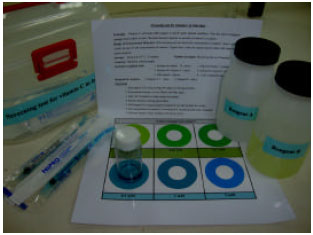
Research Article
A New Simple and Rapid Colorimetric Screening Test for Semi-qualitative Analysis of Vitamin C in Fruit Juices Based on Prussian Blue
Department of Chemistry, Faculty of Science and Technology, Rajamangala University of Technology Thanyaburi, Pathumthani 12110, Thailand
P. Chumsaeng
Department of Chemistry, Faculty of Science and Technology, Rajamangala University of Technology Thanyaburi, Pathumthani 12110, Thailand
S. Jongjinakool
Department of Chemistry, Faculty of Science and Technology, Rajamangala University of Technology Thanyaburi, Pathumthani 12110, Thailand
K. Chantu
Department of Chemistry, Faculty of Science and Technology, Rajamangala University of Technology Thanyaburi, Pathumthani 12110, Thailand
W. Nolykad
Department of Chemistry, Faculty of Science and Technology, Rajamangala University of Technology Thanyaburi, Pathumthani 12110, Thailand