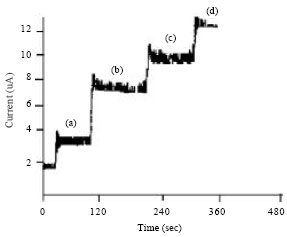
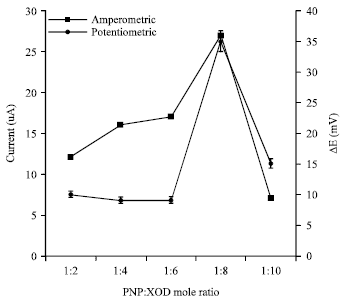
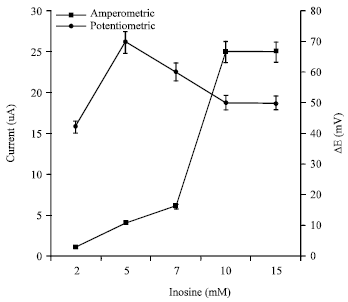
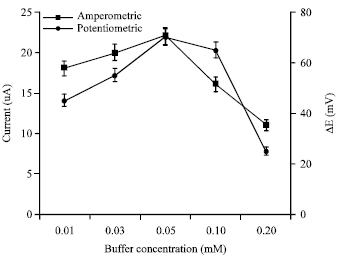
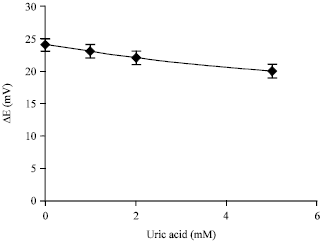
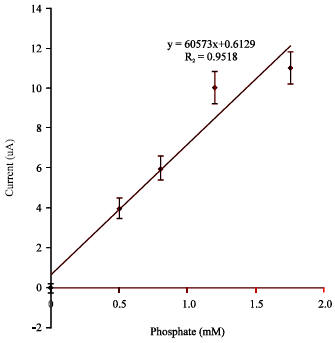
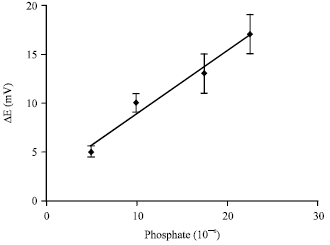
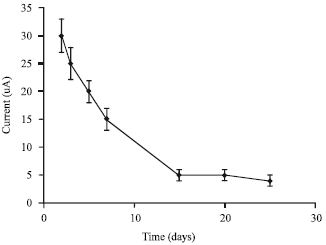
Research Article
Polypyrrole Based Amperometric and Potentiometric Phosphate Biosensors: A Comparative Study
Nano Science and Sensor Technology Research Group, School of Applied Sciences and Engineering, Monash University, Churchill, Vic-3842, Australia
Samuel B. Adeloju
Nano Science and Sensor Technology Research Group, School of Applied Sciences and Engineering, Monash University, Churchill, Vic-3842, Australia