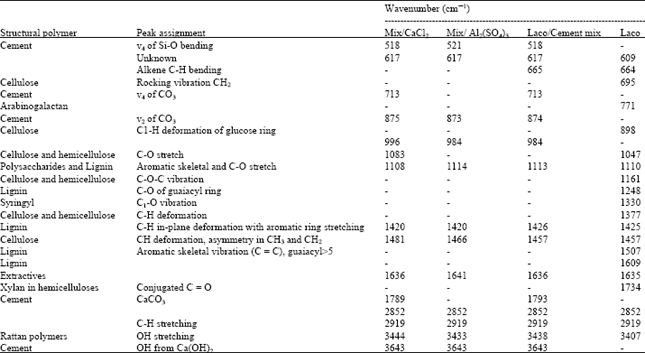
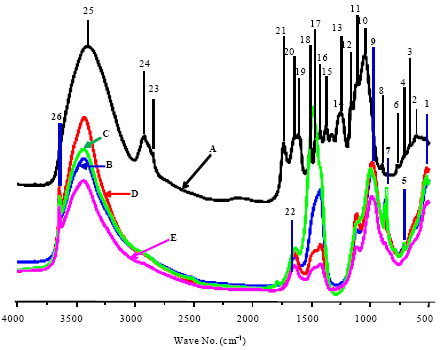
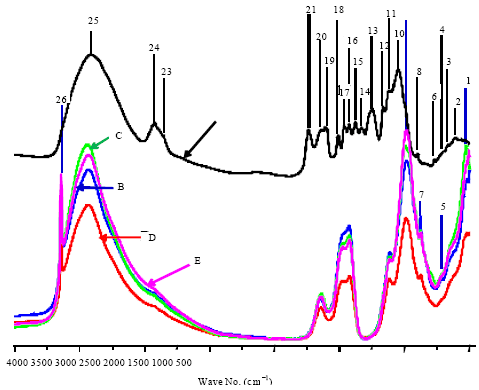
Research Article
Hydration Behaviour and Infrared Spectroscopy of Pre-treatments Effect on Portland Cement-Eremospatha macrocarpa and Laccosperma secundiflorum Systems
Department of Agricultural and Environmental Engineering, Faculty of Technology, University of Ibadan, Oyo State, Nigeria
J.S. Fabiyi
Department of Forestry and Wood Technology, Federal University of Technology, PMB 704, Akure, Ondo State, Nigeria
A.G. McDonald
Forest Products Program, College of Natural Resources, University of Idaho, Moscow, ID 83844-1132, USA