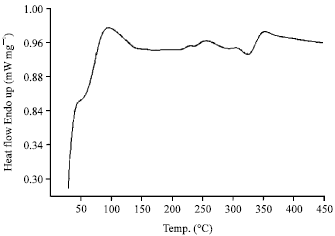
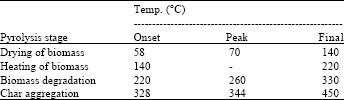
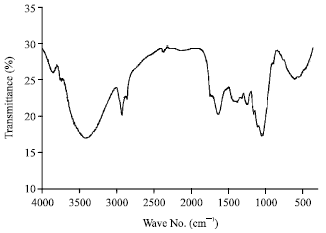
Research Article
Analysis of the Pyrolytic Fuel Properties of Empty Fruit Bunch Briquettes
Institute of Hydrogen Economy, Department of Chemical Engineering, UniversitiTeknologi Malaysia, 81310 Skudai, Johor Bahru, Malaysia
Anwar Johari
Institute of Hydrogen Economy, Department of Chemical Engineering, UniversitiTeknologi Malaysia, 81310 Skudai, Johor Bahru, Malaysia
Arshad Ahmad
Institute of Hydrogen Economy, Department of Chemical Engineering, UniversitiTeknologi Malaysia, 81310 Skudai, Johor Bahru, Malaysia