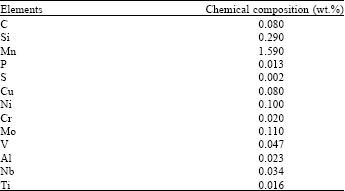
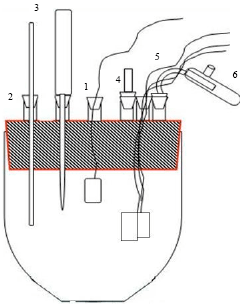
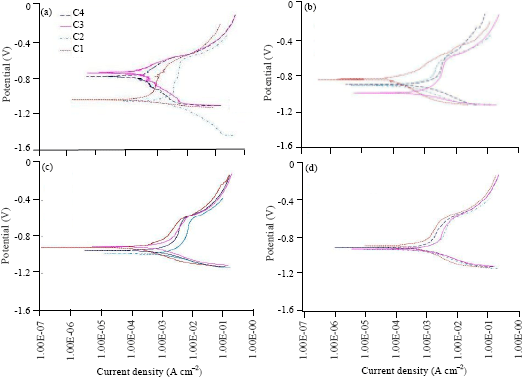
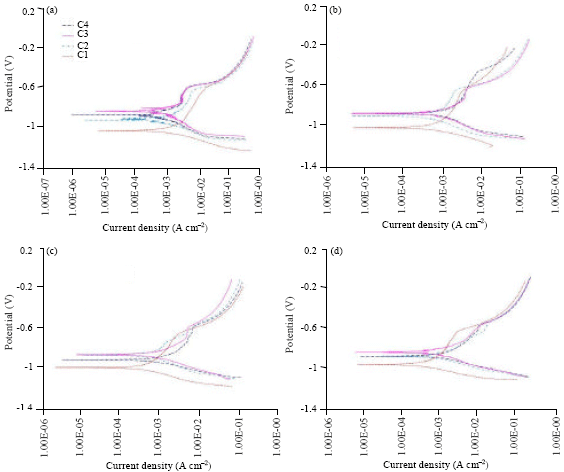
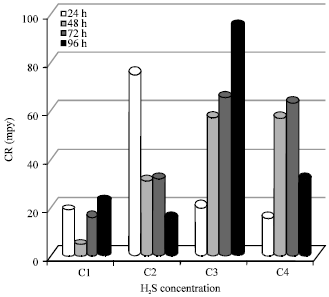
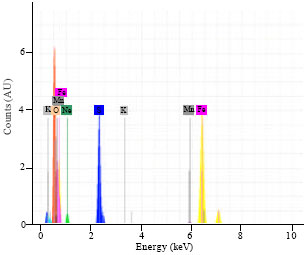
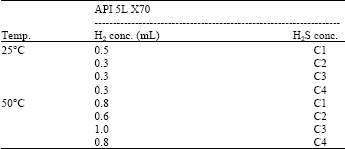
Research Article
Corrosion Investigation of Pipeline Steel in Hydrogen Sulfide Containing Solutions
Department of Mechanical Engineering, Center For Corrosion Research,Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
Hossein Taheri
Faculty of Engineering, Corrosion Engineering Group, Kish University, Kish Island, Iran
Mokhtar Che Ismail
Department of Mechanical Engineering, Center For Corrosion Research,Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
Abolghasem Dolati
Department of Materials Engineering, Sharif University of Technology, Tehran, Iran