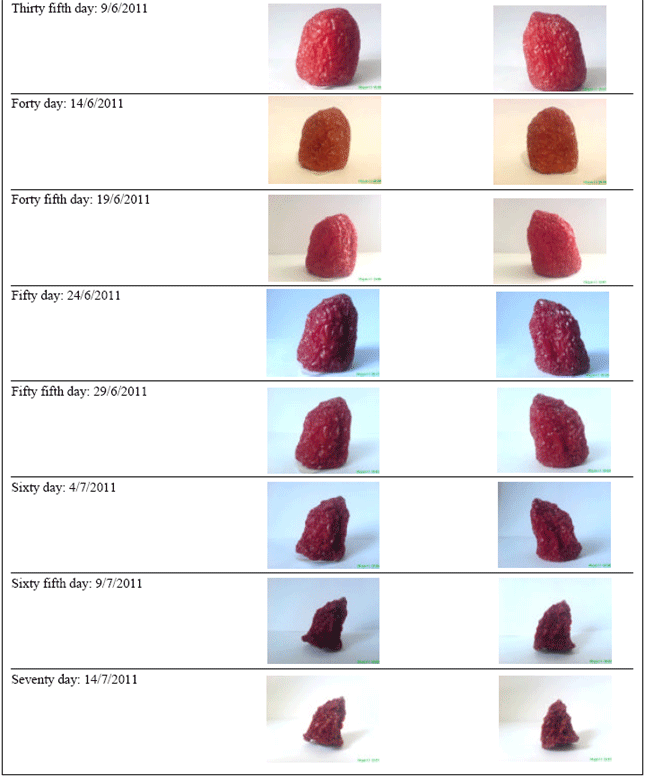
Research Article
Protection Effect of Gold Nanoparticles Coated on Fruit and Vegetables Using PVD Method
Faculty of Science, Urmia Branch, Islamic Azad University, Urmia, Iran
Sakine Shirvaliloo
Young Researches Club, Mahabad Branch, Islamic Azad University, Mahabad, Iran