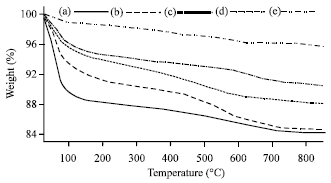
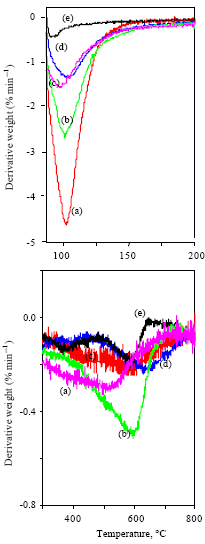
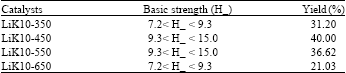
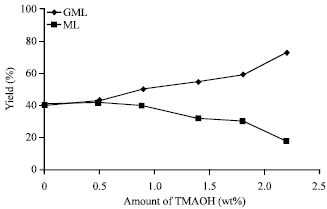
Research Article
Effect of Calcination Temperature and Tmaoh on Catalytic Activity of Basic Clay
School of Chemical Engineering, Engineering Campus, Universiti Sains Malaysia, 14300 Nibong Tebal, Pinang, Malaysia
A .Z. Abdullah
School of Chemical Engineering, Engineering Campus, Universiti Sains Malaysia, 14300 Nibong Tebal, Pinang, Malaysia
R. Zakaria
School of Chemical Engineering, Engineering Campus, Universiti Sains Malaysia, 14300 Nibong Tebal, Pinang, Malaysia