Research Article
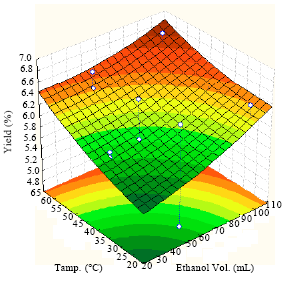
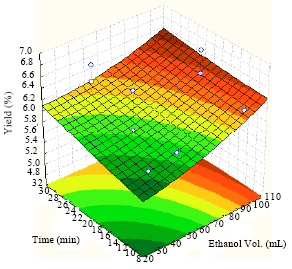
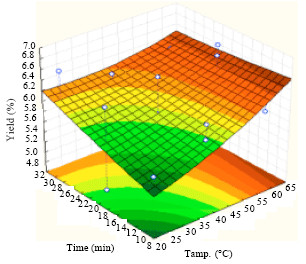
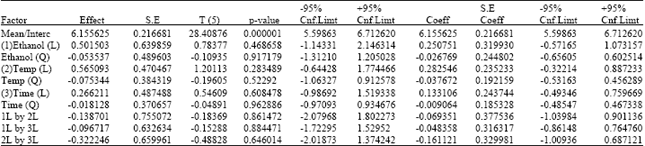
Extraction of Algae Oil from Nannocloropsis sp.: A Study of Soxhlet and Ultrasonic-Assisted Extractions
Department of Chemical Engineering, Faculty of Industrial Technology, Universitas Ahmad Dahlan, Jogjakarta, Indonesia
R. M. Yunus
Department of Chemical Engineering, Faculty of Chemical and Natural Resources Engineering, Universiti Malaysia Pahang, MEC City, 26300, Gambang, Kuantan, Pahang Darul Makmur, Malaysia
M. Mel
Department of Biotechnology Engineering, Faculty of Engineering, International Islamic University Malaysia, P.O. Box 10, 50728, Kuala Lumpur, Malaysia