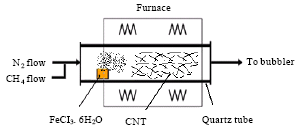
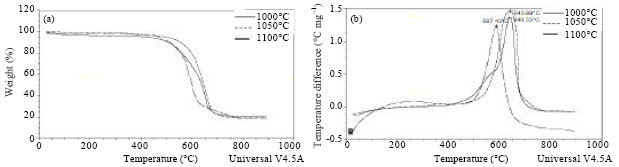
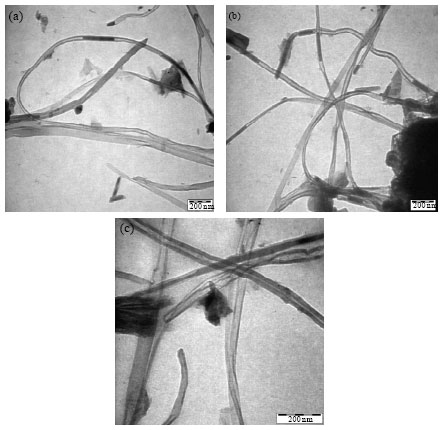
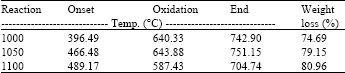
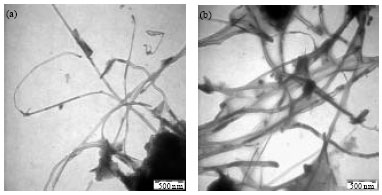
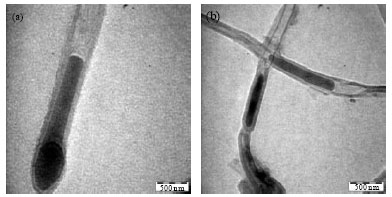
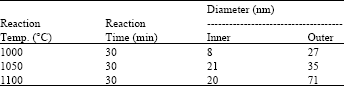Research Article
Effect of Different Parameters on the Morphology of Carbon Nanotubes Structures Grown by Floating Catalyst Method
School of Chemical Engineering, Universiti Sains Malaysia, 14300, Nibong Tebal, Pulau Pinang, Malaysia
S. P. Chai
School of Engineering, Monash University, Jalan Lagoon Selatan, 46150, Bandar Sunway, Selangor, Malaysia
S. H. Tan
School of Chemical Engineering, Universiti Sains Malaysia, 14300, Nibong Tebal, Pulau Pinang, Malaysia
A. R. Mohamed
School of Chemical Engineering, Universiti Sains Malaysia, 14300, Nibong Tebal, Pulau Pinang, Malaysia