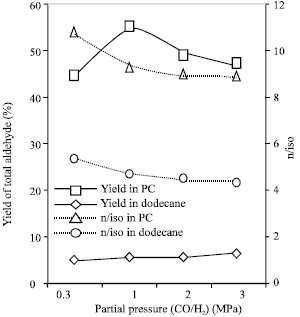
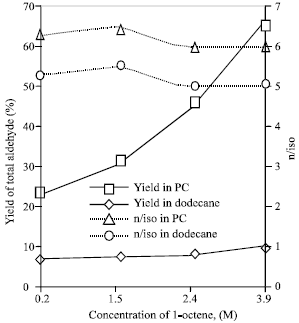
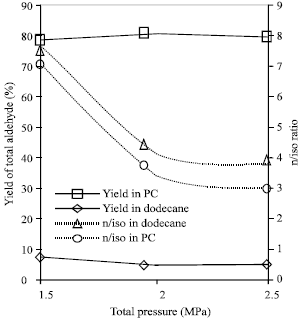
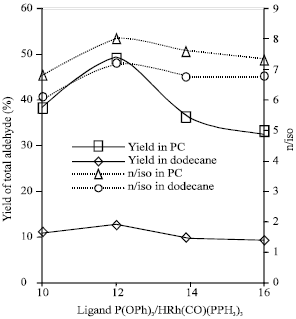
Research Article
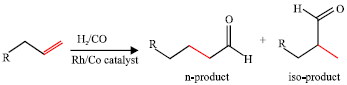
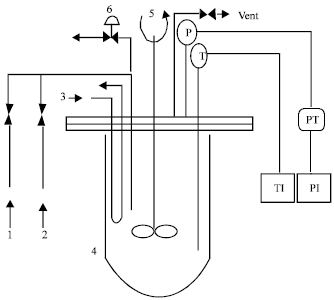
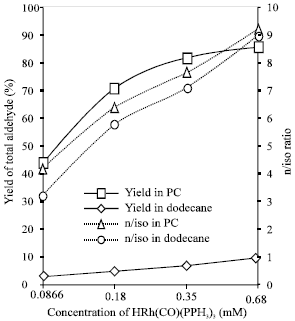
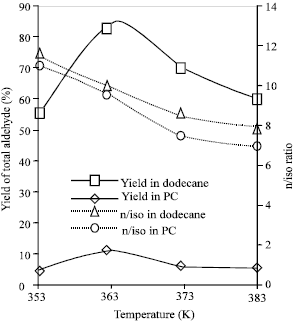
Selectivity of Rhodium-catalyzed Hydroformylation of 1-Octene in a Thermomorphic Solvent System
Department Chemical Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
Hilmi Mukhtar
Department Chemical Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
Binay K. Dutta
Department Chemical Engineering, The Petroleum Institute, Abu Dhabi, UAE