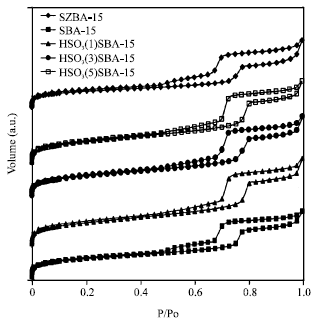
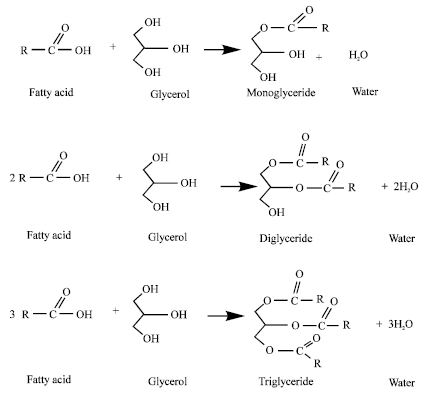
Research Article
Post Synthetically Functionalized SBA-15 with Organosulfonic Acid and Sulfated Zirconia for Esterification of Glycerol to Monoglyceride
School of Chemical Engineering, Universiti Sains Malaysia, 14300 Nibong Tebal, Pinang, Malaysia
Ahmad Zuhairi Abdullah
School of Chemical Engineering, Universiti Sains Malaysia, 14300 Nibong Tebal, Pinang, Malaysia
Abdul Rahman Mohamed
School of Chemical Engineering, Universiti Sains Malaysia, 14300 Nibong Tebal, Pinang, Malaysia