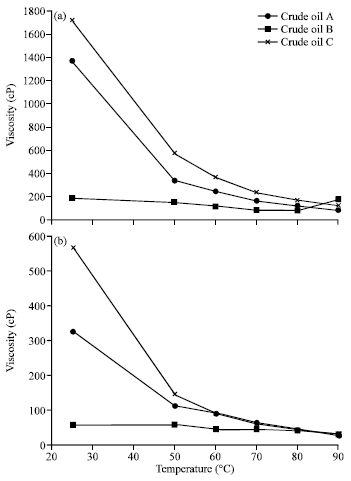
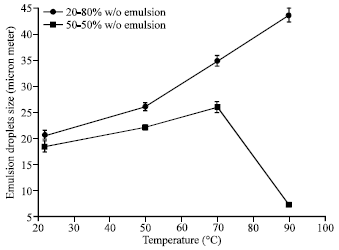
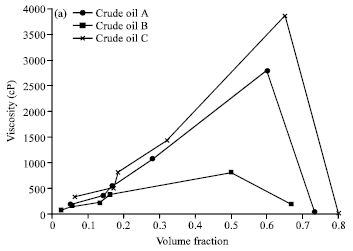
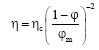Research Article
Catastrophic and Transitional Phase Inversion of Water-in-Oil Emulsion for Heavy and Light Crude Oil
Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Malaysia
Abdurahman H. Nour
Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Malaysia
Azhary H. Nour
Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Malaysia