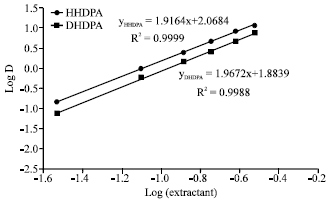
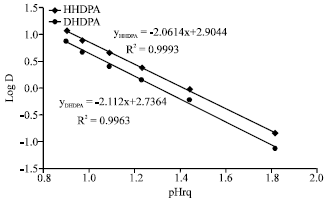
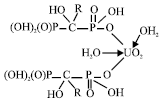
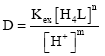Research Article
The Solvent Extraction of Uranium (VI) using Hydroxyalkylenediphosphonic Acids
Nuclear Research Center of Draria, Box 43 Sebala, Draria, Algiers, Algeria
A.Y. Badjah Hadj Ahmed
Department of Chemistry, College of Science,University of King Saud, P.O. Box 2455, Riyadh 11451, Saudi Arabia
M. Attou
Nuclear Research Center of Draria, Box 43 Sebala, Draria, Algiers, Algeria
A.H. Elias
Department of Chemistry, Faculty of Sciences, University of Mouloud Mammeri,P.O. Box 17, Tizi-Ouzou, Algeria
M.A. Didi
Laboratory of Catalysis, Department of Chemistry, University of Tlemcen, P.O. Box 119, Tlemcen, Algeria