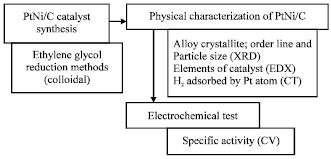
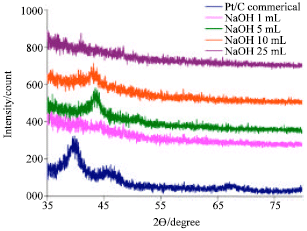
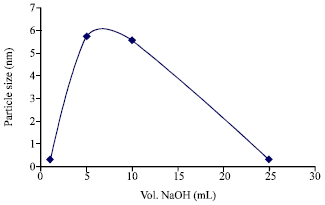
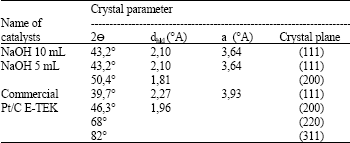
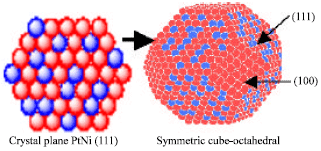
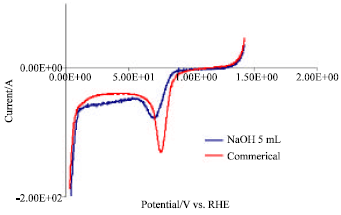
Research Article
The Effect of NaOH in The Formation PtNi/C Nanocatalyst for Cathode of PEMFC
Department of Chemical Engineering, Faculty of Engineering, University of Indonesia, Kampus UI Depok-16424, Indonesia
W. Widodo Purwanto
Department of Chemical Engineering, Faculty of Engineering, University of Indonesia, Kampus UI Depok-16424, Indonesia
Mohammad Nasikin
Department of Chemical Engineering, Faculty of Engineering, University of Indonesia, Kampus UI Depok-16424, Indonesia
Lilik Hendrajaya
Institute of Technology Bandung, Jl. Ganesha 10 Bandung 40132, Indonesia