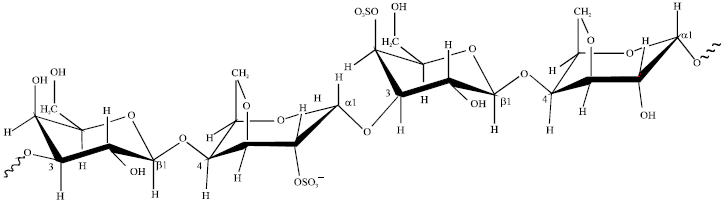
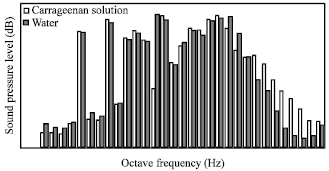
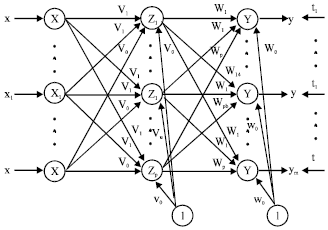
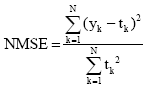Research Article
Estimation of Carrageenan Concentration by Using Ultra Sonic Waves and Back Propagation Neural Networks
Faculty of Chemical and Natural Resources Engineering, Universiti Malaysia Pahang, 25000 Kuantan, Pahang Darul Makmur, Malaysia
Duduku Krishnaiah
School of Engineering and Information Technology, Universiti Malaysia Sabah, Locked Bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
Awang Bono
School of Engineering and Information Technology, Universiti Malaysia Sabah, Locked Bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
Paulraj Pandiyan
Faculty, School of Mechatronic Engineering, Universiti Malaysia Perlis, 02600 Jejawi, Malaysia
Rosli Bin Mohd Yunus
Faculty of Chemical and Natural Resources Engineering, Universiti Malaysia Pahang, 25000 Kuantan, Pahang Darul Makmur, Malaysia
Naveena Lakshmi
Faculty of Chemical and Natural Resources Engineering, Universiti Malaysia Pahang, 25000 Kuantan, Pahang Darul Makmur, Malaysia