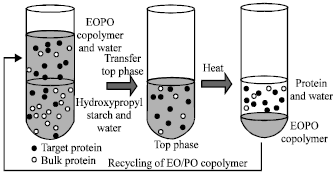
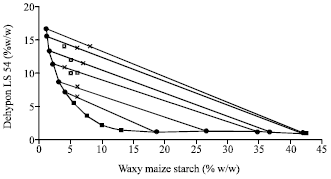
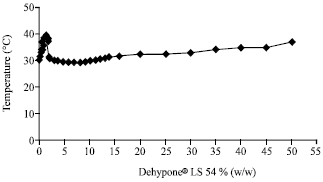
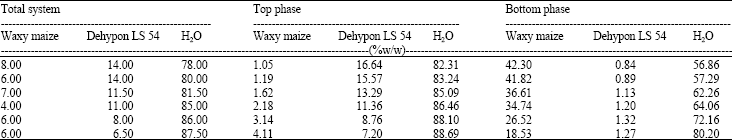
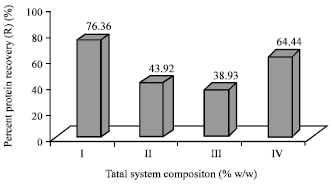
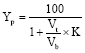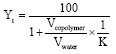Research Article
Application of the Aqueous Two-phase Thermoseparating Systems of Dehypon® LS 54-the Waxy Maize Starch for Protein Extraction
Department of Chemical and Process Engineering, Faculty of Engineering, Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia
Jamaliah Md. Jahim
Department of Chemical and Process Engineering, Faculty of Engineering, Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia