Research Article
Differential Sensitivities of Different Scenedesmus obliquus Strains to the Allelopathic Activity of the Macrophytes Stratiotes aloides
Department of Biology, Faculty of Science, King Khalid University, Abha, 9019, Saudi Arabia
Allelopathic inhibition of phytoplankton is one of the mechanisms thought to stabilize the macrophyte-dominated clear-water state in shallow eutrophic lakes (Hilt and Gross, 2008).
Absence or presence of aquatic macrophytes could be the main cause of the water quality and plankton availability variation between perennial and non-perennial ponds (Hossain et al., 2008).
It has been reported that many submerged macrophytes, such as Myriophyllum spicatum (Elisabeth et al., 1996), Ceratophyllum demersum (Korner and Nicklisch, 2002), Chara (Mulderij et al., 2003), Najas marina (Gross et al., 2003), Stratiotes aloides (Mulderij et al., 2005a, b) and Elodea (Erhard and Gross, 2006) may secrete allelochemicals to inhibit algal growth. Besdies, allelochemicals probably also interfere with many physiological functions of algal cells in terms of changes in algal morphology and biomass (Gross et al., 2003).
The submerged macrophyte Stratiotes aloides (Hydrocharitaceae) is a very common during spring and summer months in ponds in Asir region, Southwest of Saudi Arabia (Mohamed and Al-Shehri, 2010). During the submerged phase (early spring), allelopathy would be a possible mechanism to gain a competitive advantage in the struggle for light. Many studies showed the allelopathic effects of S. aloides on phytoplankton (Jasser, 1995; Van Donk and van de Bund, 2002; Mulderij et al., 2005a, b, 2006, 2007) and filamentous algae (Mulderij et al., 2009). However, phytoplankton species exhibited differential sensitivity to allelopathic activity of S. aloides. In general, more cyanobacteria than other phytoplankton species were inhibited (Mulderij et al., 2007). Most studies on the allelopathic effect of Stratiotes aloides on phytoplankton growth were conducted in the laboratory using the green alga Scenedesmus obliquus as a target organism. Scenedesmus is among the most common phytoplankton genera in freshwater ecosystems and has a worldwide distribution (Lurling, 2003) including Saudi Arabia lakes (Fathi et al., 2009; Al-Sheikh and Fathi, 2010). Therefore, this alga is expected to occur in similar habitats as macrophytes: mesotrophic to slightly eutrophic ecosystems. However, the results of these studies are controversial. Some studies reported an inhibitory effect on Scenedesmus by S. aloides (Van Donk and van de Bund 2002; Mulderij et al., 2005b), while other studies showed no effect (Jasser, 1995; Mulderij et al., 2005a). This could be due to the difference in the origin of S. obliquus strains used in these studies, as the strains originating from the same habitat of Startiotes may be less sensitive to its allelopathic activity than strains that had not been in contact with Startiotes. To prove this hypothesis, the present study investigated the allelopathic effects of S. aloides extract on the growth and antioxidative system of two strains of S. obliquus from different habitats.
Macrophyte and target organisms: Stratiotes aloides was collected in May 2008 from Tanumah pond, Southwest of Saudi Arabia. Tanumah pond is about 2000 m2 in area and located 120 km North Abha city (19 10° N, 42 04°E). The pond is found below Al-Dahnaa dam which provides the water into this pond. We used axenic cultures of Scenedesmus obliquus of different origin. Strain SZA1 was isolated from a pond not containing Stratiotes, while strain SZA2 was isolated from the same pond where the macophyte S. aloides was collected. Stratiotes plants were carefully rinsed with tap water and dried with filter papers. The plant material was cut and homogenized (10 mg mL-1) in aquoues acetone (50%) for 2 h under continuous stirring (150 rpm) according to Mulderij et al. (2007), who stated that allelopathically active compound(s) from Stratiotes are moderately lipophilic. Extracts were filtered (Whatman GF/C) to remove plant particles, evaporated under sterilized air conditions to dryness. The residue was re-suspended in 50% (v/v) methanol at a final concentration of 100 mg dry material per ml and stored at -20°C until use.
Experimental set up: Prior to the laboratory experiment, S. obliquus strains were grown in batch culture in 250 mL Erlenmeyer flasks containing 100 mL BG-11 medium at 21°C. The exponentially growing cells were used as inoculums for the start of the experiment. Each cyanobactera species was cultivated in 250 mL Erlenmeyer flasks containing 100 mL BG-11 medium which was supplemented with different concentrations (1, 10, 20, 40 mg original dry plant material/mL) of 50% methanol extract of S. aloides. Cultures without any S. aloides extract were taken as controls. Each treatment was made in triplicate. The cultures were incubated with stirring (100 rpm) at 25°C and illumination of 70 μmol m-2 sec-1. Control and toxin treated cells were harvested 7 days of incubation for growth measurement and biochemical biomarkers.
Growth measurement: The growth of control and toxin-treated cultures were estimated by measuring chlorophyll-a contents. Chlorophyll-a was determined by extracting the pellet of an aliquot of algal cells in methanol (96%) at 4°C overnight and the absorbances were read at 653 and 666 nm on UV/visible spectrophotometer (UV-1601 PC, Shimadzu Corporation, Kyoto, Japan). The amount of these pigments was calculated according to the formulas of Lorenzen (1967).
Lipid-peroxidation analysis: Lipid peroxidation was measured as the amount of malondialdehyde (MDA). MDA concentrations were determined as described by Heath and Packer (1968) and Shiu and Lee (2005). Briefly, treated and control algal cells were collected from culture medium by centrifugation at 1500x g for 10 min and then homogenized with 2 mL 10% (w/v) trichloroacetic acid (TCA). The homogenate was centrifugated at 12,000x g for 10 min and the supernatant (2 mL) was mixed with 2 mL of thiobarbituric acid (TBA) reagent (0.25% in 10% TCA) and heated for 30 min at 95°C. The mixture was cooled and centrifugated for 10 min at 6000x g. The absorbance was measured at 532 nm and corrected for non-specific absorbance at 600 nm. MDA contents were calculated using an extinction coefficient of 159.2 mM-1 cm-1.
Preparation of enzyme extracts: Algal cells were harvested by centrifugation at 2000x g for 10 min at 4°C. The cell pellets were washed with sterilized media and centrifugated again. For the determination of protein and reduced glutathione (GSH) and activities of GST and GPX, the pellets were resuspended in Phosphate Buffer Saline (PBS) (0.1 M, pH 7) and ground in a porcelain mortar. The extracts were washed in an aliquot of potassium phosphate buffer (50 mM, pH 7.5), collected and centrifuged for 20 min at 6,000 g (4°C). The supernatants were stored at 80°C until use. For the determination of alkaline phosphatase (APA), the pellets were ground in ice bath and extracted with 0.1 M Tris-HCl (pH 7.4) Berman (1970). The activity was estimated using the reaction mixture: 0.3 M p-Nitrophenyl phosphate and crude enzyme.
Enzymatic and non-enzymatic antioxidant assays: The protein concentrations were determined according to Bradford (1976) using bovine serum albumin (BSA) as a standard. The GSH content was measured according to the method of Griffith (1980). The GST activity was determined by measuring the conjugation of GSH and 1-chloro-2, 4 dinitrobenzene (CDNB) at 340 nm (Habig and Jakoby, 1981). The amount of enzyme producing 1 nmole product/min mg protein was defined as one unit using a molar extinction coefficient of 9.6/mM/cm. The GPX activity was measured colorimetrically using hydrogen peroxide as model substrate according to Peters and Livingstone (1996). The enzyme activity of GPX was expressed as nmole of NADPH consumed min-1 mg-1 protein based on an extinction coefficient of 6.22/mM/cm.
Statistical analysis: A parametric one-way Analysis of Variance (ANOVA) followed by Tukey multiple comparison test was used to test any significant difference in growth and antioxidant parameters between treated and control cultures of algae. The results were considered significant at the level of p<0.05. The statistical analyses were performed using the software called Statistical Package for Social Sciences (SPSS 9 for Windows, SPSS Inc., USA).
Effect of S. aloides extract on the growth of Scenedesmus strains: Effects of S. aloides extract on the production of chlorophyll-a by S. obliquus is given in Fig. 1. Chlorophyll-a concentration of SZA1 strain decreased significantly (F = 290, df = 4, p<0.05) with increasing the concentrations of Startiotes extract, while chlorophyll a production by SZA2 strain did not change significantly between treated and control cultures (F = 1.37, df = 4, p = 0.3) (Fig. 1).
Effects of S. aloides extract on protein, GSH and MDA contents: Protein contents of Scenedesmus obliquus have significantly affected (p<0.05) by S. aloides extract (Fig. 2). SZA1 showed a remarked reduction in protein contents of treated cultures with different extract concentrations compared to control (F = 456, df = 4, p<0.05) and that reduction increased with increasing the extract concentration (Fig. 2). While SZA2 did not show any significant change in protein contents between treated and control cultures (p = 0.2).
To examine whether S. aloides extract caused an oxidative stress on Scenedesmus strains, lipid peroxidation was quantified. The results showed that MDA, an indicator of lipid peroxidation has been affected differently between the two strains (there was a significant difference (p<0.05) in the amount of MDA between two strains). Increasing in the concentration of extract increased the value of MDA by SZA1 (p< 0.05), whereas there is no statistically variation in MDA contents between treated and control cultures of strain SZA2 (F = 0.43, df = 4, p = 0.8) (Fig. 3). The contents of GSH, a non-enzymatic antioxidant, decreased remarkably below the control with increasing plant extract concentration (F = 46.5, df = 4, p<0.05) in strain SZA1 (Fig. 4). The GSH contents did not differ significantly between treated and control cultures of the other strain (F = 1.3, df = 4, p = 0.3).
Effects of S. aloides extract on the activities of phosphatase and enzymatic antioxidants: S. aloides extract reduced sharply APA activity of Scenedesmus obliquus strain (SZA1) isolated from a Stratiotes-not containing pond (F = 1035.2, df = 4, p>0.05), while no significant difference in APA activity was found between treated and control cultures of the other strain (SZA2) isolated from the same habitat of Stratiotes (F = 2.7, df = 4, p = 0.1) (Fig. 5).
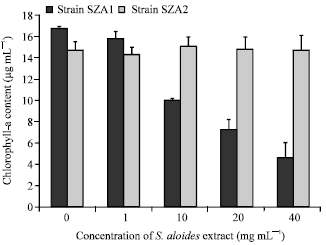 | |
| Fig. 1: | Effect of aqueous acetone extract of Stratiotes aloides on the amount of chlorophyll-a content of Scendesmus obliquus |
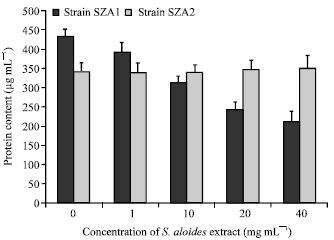 | |
| Fig. 2: | Effect of aqueous acetone extract of Stratiotes aloides on protein contents of Scendesmus obliquus |
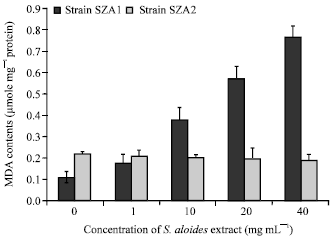 | |
| Fig. 3: | Changes in lipid perooxidation (MDA concentrations) in Scenedesmus obliquus |
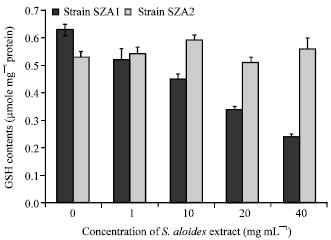 | |
| Fig. 4: | Effect of aqueous acetone extract of Stratiotes aloides on GSH contents of Scendesmus obliquus |
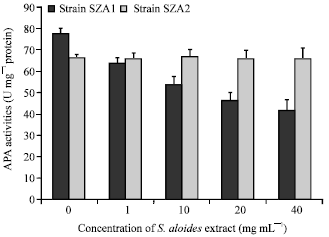 | |
| Fig. 5: | Effect of aqueous acetone extract of Stratiotes aloides on Alkaline phosphatase activity (APA) of Scendesmus obliquus |
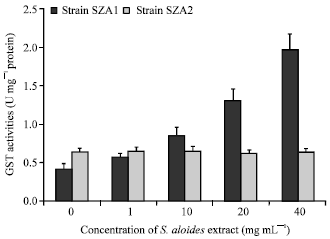 | |
| Fig. 6: | Effect of aqueous acetone extract of Stratiotes aloides on the activity of Glutathione-S-transferase (GST) activity of Scendesmus obliquus |
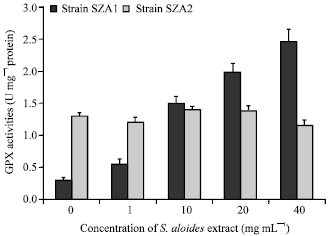 | |
| Fig. 7: | Effect of aqueous acetone extract of Stratiotes aloides on the activity of Glutathione peroxidase (GPX) of Scendesmus obliquus |
The effect of S. aloides extract on activities of antioxidative enzymes, GST and GPX varied greatly between the two strains of S. obliquus. Both GST and GPX activities increased markedly (F = 698, 181, df = 4, p<0.05, respectively) over the control with increasing macrophyte extract concentrations in treated cultures of strain SZA1, (Fig. 6, 7). On the other hand, the activities of these two enzymes did not change significantly between control and treated cultures of the other strain isolated from the same habitat of Stratiotes (F = 0.87, 1.6, p = 0.05, 0.24, respectively) (Fig. 6, 7).
The results of present study showed that aqueous acetone extract of the submerge macrophyte S. aloides inhibited the chlorophylla production by SZA1 originating from a habitat not containing Stratiotes, whereas pigment value of SZA2 from the same habitat of Stratiotes. was not significantly affected. Present results are in agreement with those of previous studies reporting the growth inhibition of strains of S. obliquus not isolated from the same habitat of Stratiotes by extracts or exudates of this macrophyte (Mulderij et al., 2005b, 2007). However, other studies reported no inhibition effect of macrophytes including Stratiotes on the growth of S. obliquus (Jasser, 1995; Mulderij et al., 2003). The discrepancy in the results of these studies could be due to the difference in the origin of these strains. The present study proved that S. obliquus strain isolated from the same habitat of Stratiotes was less sensitive to the allelopathic effect of this macrophyte than strain that had not been in contact with in contact with Startiotes. This could be due to that Scendesmus strain from the same habitat of Stratiotes has developed resistance against allelopathic substances of this macrophyte by co-evolution (Reigose et al., 1999). Previous studies described that epiphytic algal species are less sensitive or not inhibited at all than planktonic species by macrophyte allelochemiclas (Hilt, 2006; Erhard and Gross, 2006).
In the present study, the aqueous acetone extract of S. aloides decreased the APA activity of strain SZA1 of Scendesmus which had not been in contact with Stratiotes, while no significant effect was observed on APA activities of strain SZA2 isolated from the same habitat of Stratiotes. These results indicate that the inhibition of extracellular alkaline phosphatase (APA) could be one mode of action of allelopathically active compounds as proposed by Elisabeth et al. (1996). Present results also showed that APA activities were concomitant with the growth of algal cultures of tested strains exposed to different concentration of Stratiotes extract. These results supported the findings of obtained in previous studies reporting the parallel inhibition of both growth and APA activities by macrophyte allelochemicals (Elisabeth et al., 1996; Hilt, 2006).
In addition to the growth inhibition, the aqueous acetone extract of S. aloides stimulated the activity of antioxidant enzymes (GST and GPX) and increased MDA content of S. obliquus strain SZA1 isolated from a habitat not containing Stratiotes. The enhancement of the activities of antioxidant enzymes and the decrease in non-enzymatic antioxidant (GSH) indicated that S. stratiotes extract created oxidative stress condition led to an increased antioxidant capability of SZA1 strain. These results coincide with those of previous studies showing the induction of the cellular responses of antioxidant enzymes and non-enzymatic antioxidants of algal cell by macrophyte allelochemicals (Li and Hu, 2005; Singh et al., 2006; Hong et al., 2009). In addition, present results support the hypothesis that algae may respond by a burst of Reactive Oxygen Species (ROS) under stress conditions (Kupper et al., 2001; Choo et al., 2004; Wu et al., 2007). However, the increase in the activity of antioxidative enzymes might not match the production of ROS, thus resulting in an increased lipid peroxidation in algal cells (Fargaova et al., 1999; Wu et al., 2007) and leading to reduction in algal cell proliferation and cell pigments (Nagalakshmi and Prasad, 1998; Hong et al., 2009). This could explain the association of the decrease in the growth of strain SZA1 with the increase in lipid peroxidation and antioxidant enzymes (GST, GPX) activities and decrease in reduced GSH as exposed to Stratiotes extract during the present study. In contrast to strain SZA1, Strain SZA2 isolated from the same habitat of Stratiotes did not show any significant difference in lipid peroxidation, GSH contents or antioxidant enzymes activities between treated and control cultures. These results point out that the cells of this strain were not under oxidative stress and thereby their growth was not affected by S. aloides extract.
The results of present study showed that strains of the green alga S. obliquus of different origin respond differently to the allelopathic activity of the aqueous acetone extract of the macrophyte S. aloides. The macrophyte extract inhibited the growth and triggered the production of ROS in strain SZA1 isolated from Stratiotes-not containing habitat, while it had no significant effect on the growth or the antioxidative system of strain SZA2 isolated from the same habitat of Stratiotes. To my knowledge, this is the first study to compare the effect of macrophyte allelopathy on strains of the same species of green algae with different origin. The study suggests algal strains co-occurring with macrophytes in the same habitat could adapt and develop resistance against allelopathic substances of the macrophytes by co-evolution.