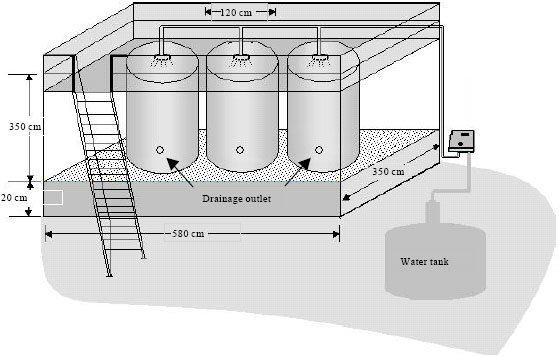
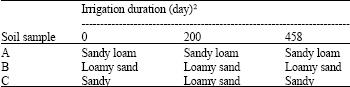
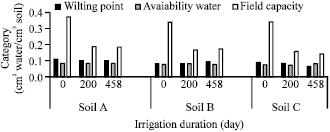
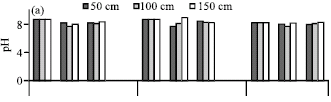
Research Article
Effect of Treated Domestic Wastewater on Physical and Chemical Characteristics of Soils
Department of Agricultural Engineering, College of Food and Agricultural Sciences, King Saud University, P.O. Box 2460, Riyadh, 11451, Kingdom of Saudi Arabia