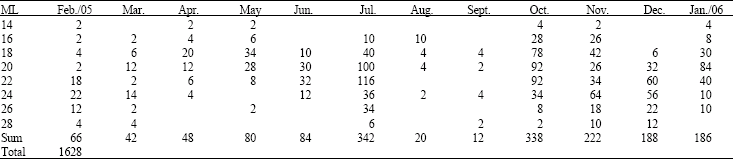
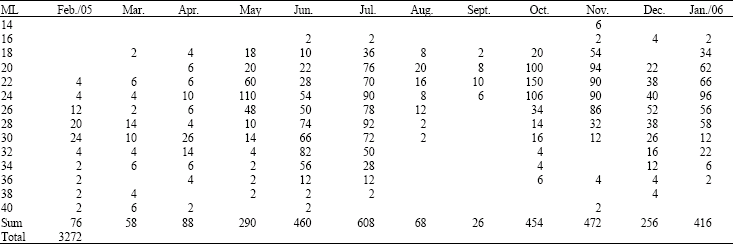
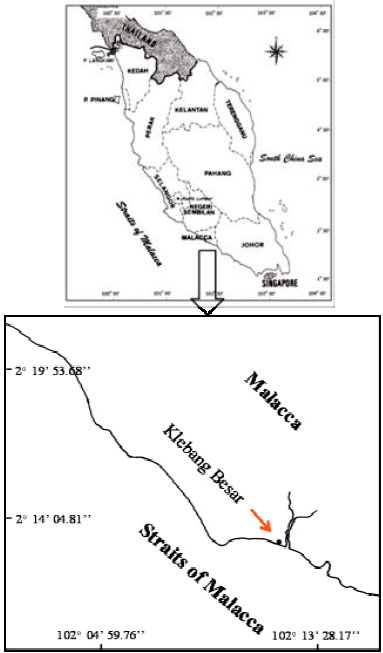
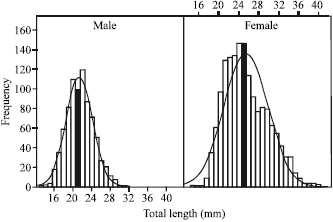
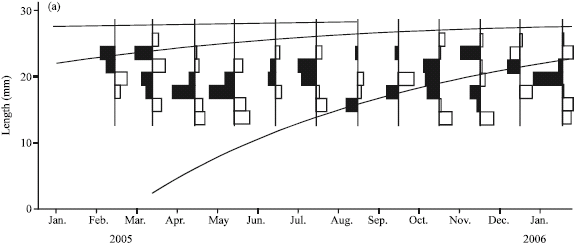
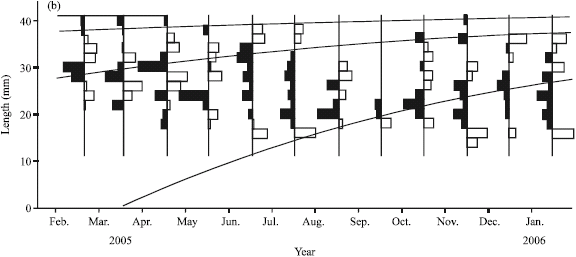
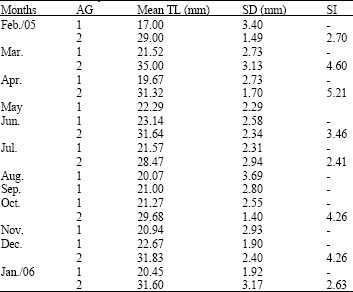
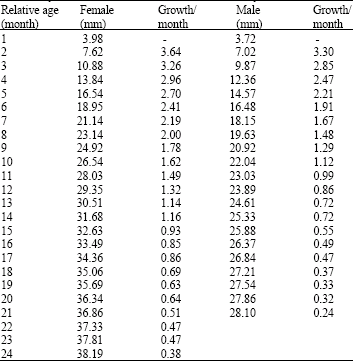
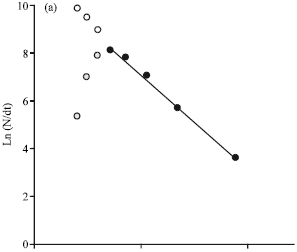
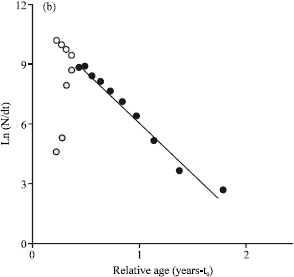
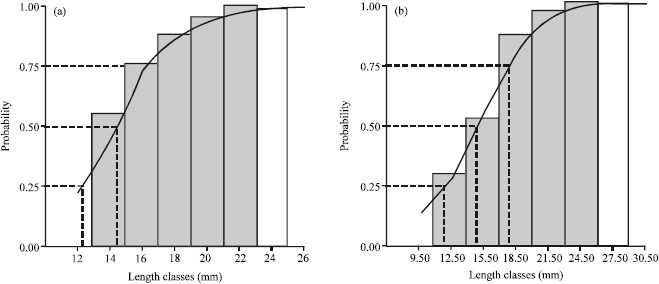
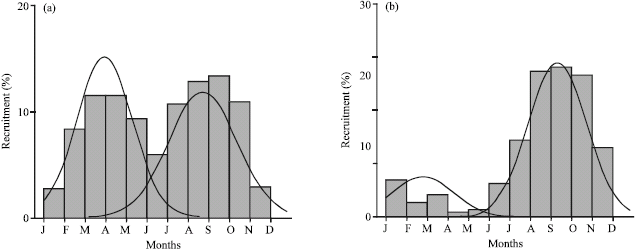
Research Article
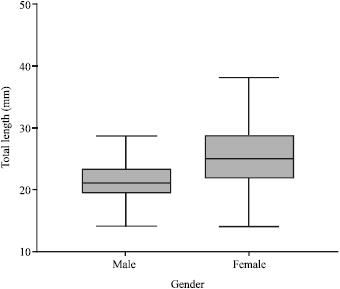
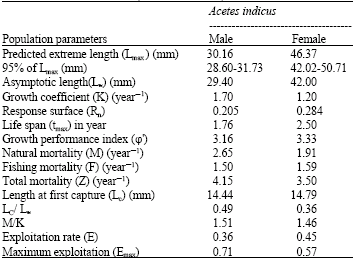
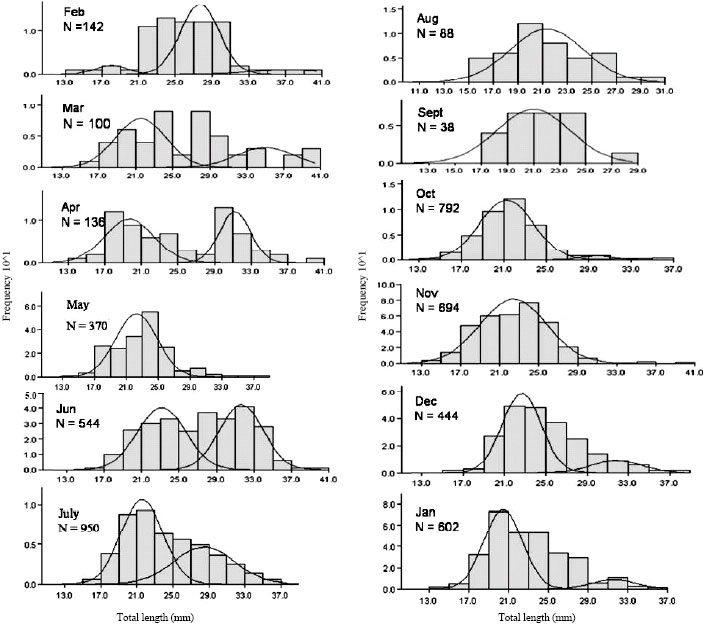
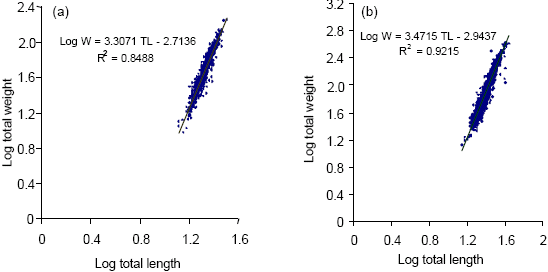
Age Structure, Growth, Mortality and Yield-Per-Recruit of Sergestid Shrimp, Acetes indicus (Decapoda: Sergestidae) From the Coastal Waters of Malacca, Peninsular Malaysia
Institute of Marine Sciences and Fisheries, University of Chittagong, Chittagong-4331, Bangladesh
A. Arshad
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
J.S. Bujang
Department of Animal Science and Fishery, Faculty of Agriculture and Food Sciences, Universiti Putra Malaysia, Bintulu Campus, 89007 Bintulu Sarawak, Malaysia
S.S. Siraj
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia