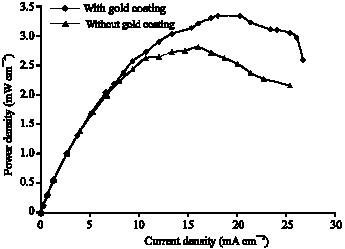
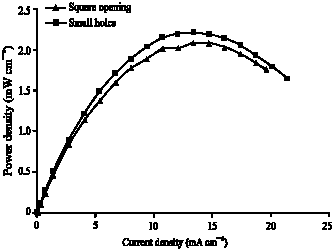
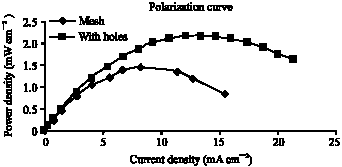
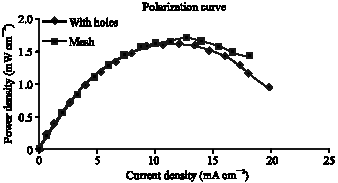
Research Article
Performance Evaluation of a Passive Direct Methanol Fuel Cell
Fuel Cell Institute,Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia
S.K. Kamarudin
Fuel Cell Institute,Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia
W.R.W. Daud
Fuel Cell Institute,Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia
Z. Yaakub
Department of Chemical and Process Engineering, Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia