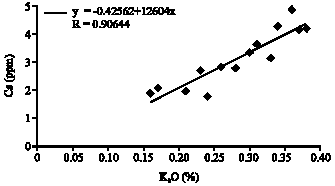
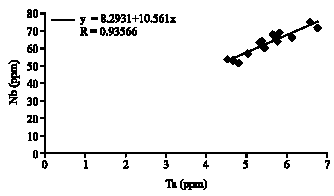
Research Article
Variability of Clays from Gounioube Deposit (Ivory Coast)
Laboratoire de Chimie des Materiaux Inorganiques, Universite de Cocody, 22 BP 582 Abidjan 22, Cote d’Ivoire
A. Abba Toure
Laboratoire de Chimie des Materiaux Inorganiques, Universite de Cocody, 22 BP 582 Abidjan 22, Cote d’Ivoire
G. Kra
Laboratoire de Chimie des Materiaux Inorganiques, Universite de Cocody, 22 BP 582 Abidjan 22, Cote d’Ivoire
J. Yvon
Laboratoire Environnement Mineralurgie, UMR CNRS-INPL 7569 Nancy Universite, 15 Avenue du Charmois, BP 40 F-54501, Vandoeuvre lès Nancy Cedex, France