Research Article
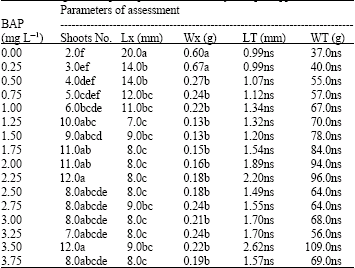
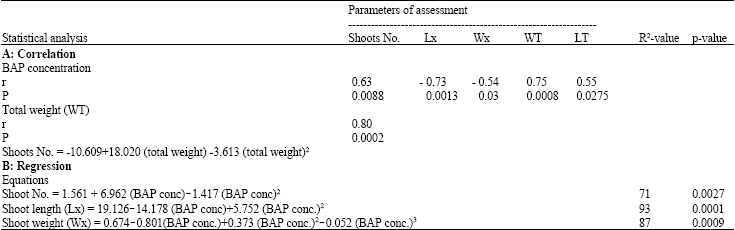
Effect of Benzylaminopurine (BAP) on in vitro Proliferation and Growth of Pineapple (Ananas comosus L. Merr.) cv. Smooth Cayenne
Institute of Biological Science, Faculty of Science, University of Malaya, 50603, Kuala Lumpur, Malaysia
R.M. Taha
Institute of Biological Science, Faculty of Science, University of Malaya, 50603, Kuala Lumpur, Malaysia