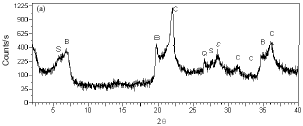
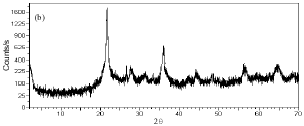
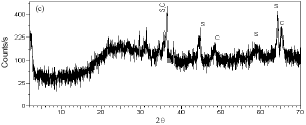
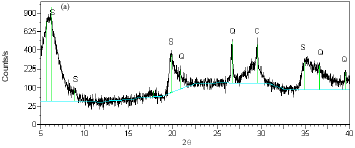
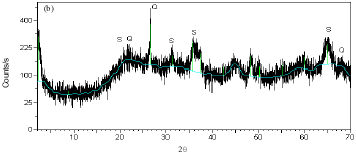
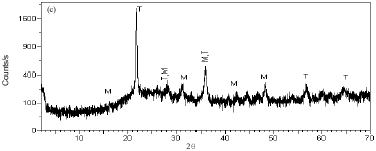
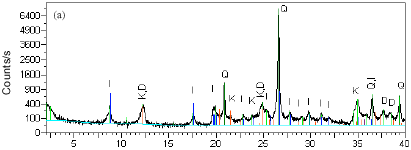
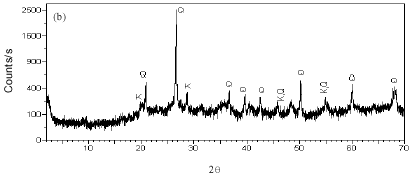
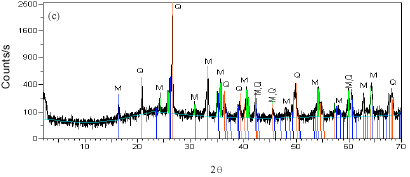
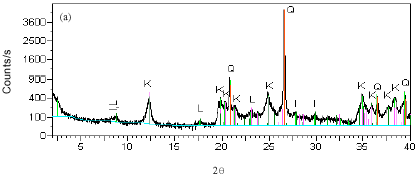
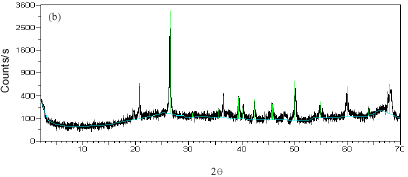
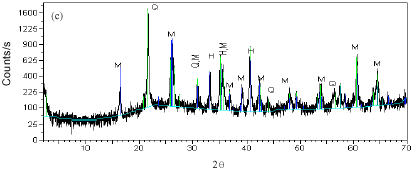
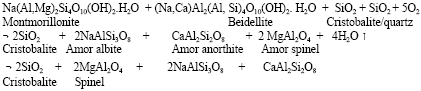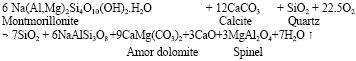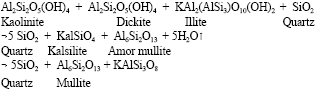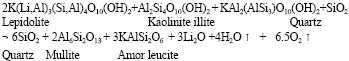Research Article
Mineral Thermochemistry of Bentonite and Kaolin Related to Their Possible Application in the Ceramic Industry
Geology, Mining and Minerals Programs, School of Physical and Mineral Sciences, University of Limpopo, P/Bag X1106, Sovenga 0727, South Africa
Antoine F. Mulaba-Bafibiandi
Department of Extraction Metallurgy, Faculty of Engineering and the Built Environment, University of Johannesburg, P.O. Box 526, Wits 2050, Johannesburg, South Africa