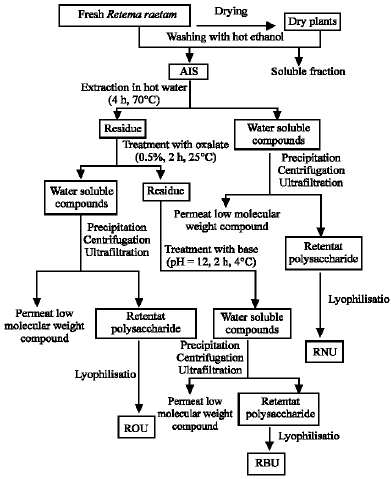
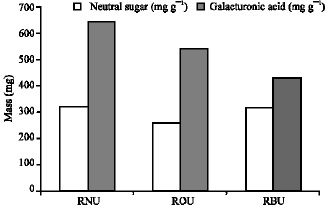
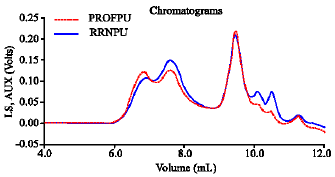
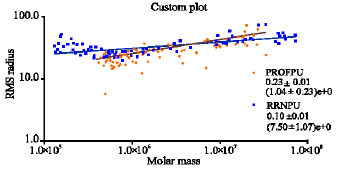
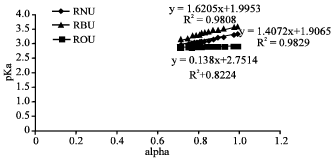
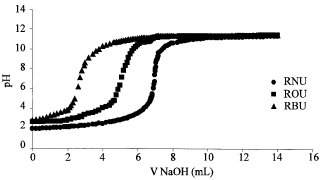
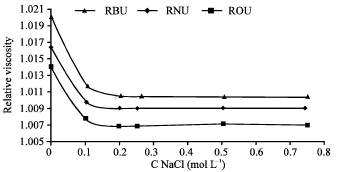
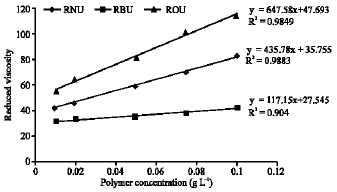
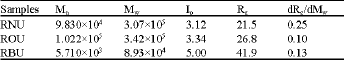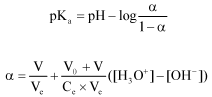Research Article
Physicochemical Properties of Pectin from Retama raetam Obtained using Sequential Extraction
Laboratoire Polym�res, Biopolym�res et Materiaux Organiques,Faculte des Sciences de Monastir, 5000 Monastir, Tunisia
H. Majdoub
Laboratoire Polym�res, Biopolym�res et Materiaux Organiques,Faculte des Sciences de Monastir, 5000 Monastir, Tunisia
S. Roudesli
Laboratoire Polym�res, Biopolym�res et Materiaux Organiques,Faculte des Sciences de Monastir, 5000 Monastir, Tunisia