Research Article
Carboxymethylation of Cassava Starch in Different Solvents and Solvent-Water Mixtures: Optimization of Reaction Conditions
Department of Chemistry, University of Ibadan, Ibadan, Nigeria
Gabiel B. Ogunmola
Institute of Genetic Chemistry and Laboratory Medicine, Bodija, Ibadan, Nigeria









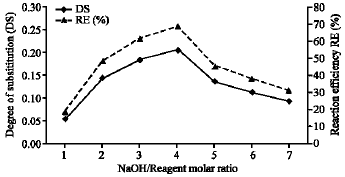
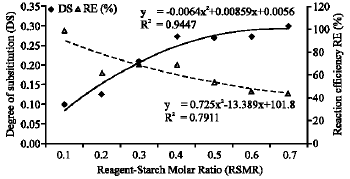
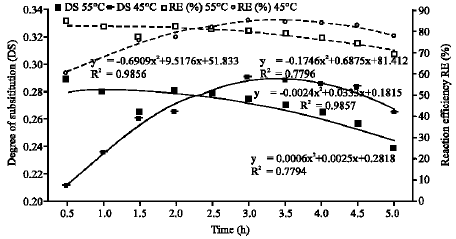
Joseph Saturday Pius Reply
Pls, Kindly help me on Article on Carboxymethylation of Maize Starch