ABSTRACT
The present study examined the preventive influences of an aqueous extract of cinnamon on carbon tetrachloride-induced some physiological alterations in the frog, Rana ridibunda. The experimental animals were divided into five batches. The first batch was untreated and served as control. The other batches were treated for 6 weeks with carbon tetrachloride, cinnamon extract plus carbon tetrachloride, cinnamon and corn oil, respectively. Haematological, biochemical and hepatosomatic index indices were chosen as physiological indicators. These parameters were evaluated at 2, 4 and 6 weeks. In comparison with control and cinnamon plus CCl4 batches, significant decreases of red blood cell count, haemoglobin concentration, haematocrit, mean corpuscular haemoglobin concentration and increases of glutamic pyruvic acid transaminase values were noted in CCl4-exposed batch at all experimental periods. Also, glutamic oxaloacetic acid transaminase and hepatosomatic index levels were significantly elevated, while mean corpuscular haemoglobin values were decreased at second and last periods. Mean cell volume values were only increased at the first period. In comparison with control batch, significant decreases of red blood cell count, haemoglobin concentration, haematocrit, and increases of glutamic oxaloacetic acid transaminase, glutamic pyruvic acid transaminase and hepatosomatic index values were observed in frogs treated with cinnamon plus CCl4 at 2 and 6 weeks. Mean cell volume and mean corpuscular haemoglobin values were statistically elevated at second period. Mean corpuscular haemoglobin concentration values were declined at last period. Moreover, the percentage changes of these parameters in cinnamon plus CCl4 batch tended to be lower than CCl4 treated the experimental animals. In addition, it is conceivable therefore, that the cinnamon aqueous extract exhibits a protective influence against carbon tetrachloride-induced some physiological changes, probably mediated through different pathways.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2007.79.90
URL: https://scialert.net/abstract/?doi=jas.2007.79.90
INTRODUCTION
Carbon tetrachloride (tetrachloromethane, CCl4) is a highly toxic chemical agent. Carbon tetrachloride is a clear oily liquid at room temperature with an aromatic sweet odor. Carbon tetrachloride is until the mid 1960's was used in fire extinguishers. Other uses, which have been discontinued, include a solvent for cleaning and household products, as a fumigant for insects and as an oral treatment for certain parasitic infections. Industrial uses include the production of chlorofluorocarbons used in refrigeration and as a degreasing solvent. These uses are expected to decline (ATSDR, 1989). CCl4 is relatively stable in the environment. Volatilization is the primary removal mechanism from water and soil. It eventually diffuses into the stratosphere where it undergoes photolysis by ultraviolet light (U.S. EPA, 1989). It can form explosive, impact-sensitive mixtures with particulates of metals including aluminum, barium, beryllium, potassium, lithium, sodium and zinc. Carbon tetrachloride also forms explosive mixtures with chlorine trifluoride, calcium hypochlorite, calcium disilicide, triethyl- dialuminum trichloride, decaborane and dinitrogen tetraoxide. It will react violently with fluorine, boranes, allyl alcohol and other related chemicals. Phosgene and Cl¯ are decomposition products when carbon tetrachloride is heated (Sax and Lewis, 1989). CCl4 is a known hepatotoxic and nephrotoxic agent (Dogukan et al., 2003; Ozturk et al., 2003; Satturwar et al., 2003; Das et al., 2004; Valcheva-Kuzmanova et al., 2004; Ha et al., 2005; Jiko et al., 2005; Shailajan et al., 2005). Also, the experimental studies showed that CCl4-induced several physiological alterations (Mortiz and Pankow, 1989; MacDonald-Wicks and Garg, 2002; Taira et al., 2004; Hui et al., 2006). Quantitative studies on the distribution of carbon tetrachloride in humans were not available, but animal studies indicate that the compound is generally distributed as a function of blood flow and fat content of the tissues. Using an autoradiographic technique and an inhalation protocol, Bergman (1983) demonstrated a considerable higher uptake of [14C] carbon tetrachloride into the white matter of brain, spinal cord and spinal nerves of rats than in the kidney, lung, spleen, muscle and blood. A similar distribution was reported following oral exposure in rats, except a higher concentration was found in the liver than in the brain. This is due to the absorption of the material from the intestine into the portal circulation. Peak concentrations in the blood, striated muscle, brain and liver were reached within 2 h after exposure but were not reached in the fat tissue until 5.5 h. Maximum concentrations in the fat were 50 times the peak blood concentrations (Marchand et al., 1970; ATSDR, 1989).
Humans are sensitive to carbon tetrachloride intoxication by oral, inhalation and dermal routes. Oral and inhalation exposure to high concentrations of carbon tetrachloride results in acute central nervous system effects including dizziness, vertigo, headache, depression, confusion, in coordination and, in severe cases, respiratory failure, coma and death. Gastrointestinal problems including nausea, abdominal pain and diarrhea, often accompany these narcotic effects. Liver and kidney damage can appear after the acute symptoms subside. All symptoms can occur following a single oral or inhalation exposure. Milder narcotic effects followed by liver and kidney damage have been reported following dermal exposure. Although an inhalation exposure of about 1000 ppm for a few minutes to hours will cause the narcotic effects in 100% of the population, large variations in sensitivity are seen. Alcohol intake greatly increases human sensitivity to carbon tetrachloride; consequently, exposure to 250 ppm for 15 min can be life threatening to an alcoholic. Subchronic and chronic exposure to doses as low as 10 ppm can result in liver and kidney damage (ATSDR, 1989; Sax and Lewis, 1989). Lung damage has also been reported in animals and humans but is not route specific and is believed to be secondary to kidney damage (Sax and Lewis, 1989). Prolonged exposure has been observed to cause visual effects in both humans and animals. Changes in the visual field, reduced corneal sensitivity, subnormal dark adaption and changes in color perception have been reported in humans exposed by inhalation to a minimum concentration of 6.4 ppm, 1 h/day for an average of 7.7 years. Increased hepatic enzyme activities indicative of liver damage have also been observed (Smyth et al., 1936; Barnes and Jones, 1967; Moeller, 1973; ATSDR, 1989). Maternal toxicity and fetotoxic effects have been reported in rats following oral or inhalation exposure to carbon tetrachloride during gestation (Wilson, 1954; Schwetz et al., 1974). Repeated inhalation exposure of male rats to carbon tetrachloride concentrations of 200 ppm or greater has been reported to cause degeneration of the testicular germinal epithelium as well as severe liver and kidney damage (Adams et al., 1952). A subchronic reference dose (RfDs) of 0.007 mg/kg/day has been calculated for oral exposure from a no-observed-adverse-effect level (NOAEL) of 0.71 mg/kg/day determined in a 12 weeks rat study (U.S. EPA, 1992a). Significantly higher doses caused minimal liver damage (Bruckner et al., 1986). A dose of 7.1 mg/kg/day was considered a lowest-observed-adverse-effect level (LOAEL). A chronic reference dose (RfDc) of 0.0007 mg/kg/day was calculated by adding an additional uncertainty factor of 10 to account for the use of a subchronic study. Confidence in the oral RfD values is rated medium by U.S. EPA (1992b). A chronic or subchronic reference concentration (RfC) for inhalation exposure is currently under development by the U.S. EPA. Although data for the carcinogenicity of carbon tetrachloride in humans are inconclusive, there is ample evidence in animals that the chemical can cause liver cancer. Hepatocellular carcinomas have been induced in hamsters, rats and mice after oral carbon tetrachloride treatment for 16 to 76 weeks. Liver tumors have also been demonstrated in rats following inhalation exposure, but the doses were not quantitatively established. The U.S. EPA weight-of-evidence classification for both oral and inhalation exposure is B2, probable human carcinogen based on adequate animal evidence. Carcinogenicity slope factors of 0.13 (mg/kg/day) for oral exposure and 0.053 (mg/kg/day) for inhalation exposure have been calculated from the oral exposure experiments with hamsters, rats and mice (Edwards and Dalton, 1942; Della Porta et al., 1961; NCI, 1976a, b; Weisburger, 1977; U.S. EPA, 1992a,b). A drinking water unit risk of 3.7x10-6 (g L-1) and an inhalation unit risk of 1.5x10-5 (g m-3) have also been calculated by U.S. EPA (1992b).
Herbs are most often defined as any part of a plant that is used in the diet for its aromatic properties (Hacskaylo, 1996; Smith and Winder, 1996; Davidson, 1999). Recently, however, herbs have also been identified as sources of various phytochemicals, many of which possess important antioxidant activity (Larson, 1988; Velioglu et al., 1998; Kähkönen et al., 1999). Spices are dried herbs and condiments are spices and other flavourings added to food at the table (Hacskaylo, 1996; Smith and Winder, 1996; Davidson, 1999). Since prehistoric times, herbs have also been the basis for nearly all medicinal therapy until synthetic drugs were developed in the 19th century. Today, herbs are still found in 40% of prescription drugs (Hacskaylo, 1996; Smith and Winder, 1996; Davidson, 1999). In addition, herbs are used for many other purposes including beverages such as tea, dyeing, repellents, fragrances, cosmetics, charms, smoking and industrial uses. Also, spices have been used historically to increase metabolism, raise body heat (thermogenesis), improve digestion and assimilation and potentiate the effects of other substances. For this reason, in many herbal traditions, small amounts of hot pungent spices were added to many traditional compounds. Cinnamon is the inner bark of a tropical evergreen tree. There are many different species, between 50 and 250. The two main varieties are Cinnamomum cassia and Cinnamomum zeylanicum, Family: Lauracae. In ancient Egypt cinnamon was used medicinally and as a flavouring for beverages, It was also used in embalming, where body cavities were filled with spiced preservatives. In the ancient world cinnamon was more precious than gold. Cinnamon was known in medieval Europe, where it was a staple ingredient, along with ginger, in many recipes. In India and Europe cinnamon was traditionally taken as a warming herb for cold conditions. The herb stimulates circulation, especially in the fingers and toes. Cinnamon is also a traditional remedy for digestive problems, such as nausea, vomiting and diarrhea, as well as for aching muscles and other symptoms of viral conditions such as colds. Also, Chinese cinnamon is a warming and strengthening herb traditionally used to support conditions associated with cold, congestion and energy deficiency. Cinnamon acts as a tonic to the whole system to stimulate circulation, enhance the immune system and improve sexual health. It is used to improve circulation to the lower body in formulas that treat impotence, premature ejaculation, cold and weak legs and backache. It is frequently combined with Yin and Yang Essence tonic herbs and with Qi tonics to build energy and to fire up sexual energy. Cinnamon is also used as a relaxant to reduce anxiety and stress and relieve cramps. Cinnamon warms and stimulates the digestive system, useful in weak digestion, colic, griping, diarrhea, nausea and vomiting, wind and distension. Convalescence-Cinnamon is a supportive and strengthening herb for weak digestions. It is used specifically in the treatment of debility and in convalescence Gynecological remedy. The herb promotes menstruation and relieves menstrual pain in formulas for women. Also, Researchers administered cinnamon extract at different dosages to the experimental animals and at the end of the study, blood glucose concentration was significantly decreased in a dose-dependent manner. Serum insulin levels and HDL-cholesterol were significantly higher and the concentration of triglycerides and total cholesterol were significantly lower. These results suggest that cinnamon extract has a regulatory role in blood glucose level and lipids and it may also exert a blood glucose-suppressing effect by improving insulin sensitivity or slowing absorption of carbohydrates in the small intestine (Kim et al., 2006).
It is worth to mention that to date, no one has investigated the relationship or interaction between cinnamon and carbon tetrachloride affects. Thus the present study was conducted to examine the effect of cinnamon extract on CCl4-induced physiological alterations. Furthermore, this study may help to understand the extent of interaction between spices supplementation, cinnamon specially, in toxicants induced physiological disturbances, its prevention in animal model and allowed for preventive applying in human status.
MATERIALS AND METHODS
Animals: Adult male and female frogs (Rana ridibunda) weighing 30.73-36.48 g were collected from Al-Qatif farms, Saudi Arabia. The experimental animals were acclimated for 2 weeks in glass aquaria (50x50x80 cm). The bottoms of these glass aquaria were covered with wet sponges. They were maintained on natural day light in an ambient temperature of 23±1°C. Frogs were fed with 0.5 mL of prepared meal (50% total protein) by intragastric intubation, twice daily.
Preparation of cinnamon extract: Fine dried powder of cinnamon bark was obtained from a commercial market. Five grams of cinnamon powder were homogenized with 200 mL of distilled water. Cinnamon homogenate was carefully centrifuged and the supernatant was used as an aqueous extract for further investigation. Cinnamon extract was freshly prepared weekly, kept in a dark container and stored at 4°C.
Experimental design: Frogs were assigned to one of five batches each of 100 frogs. The first batch was untreated and served as control. Animals of second batch were received CCl4 by intragastric intubation at a dose of 5 μL in 0.1 mL corn oil/frog, five times weekly for 6 weeks. Frogs of third batch were fed with cinnamon extract by intragastric intubation at a dose of 0.5 mL/frog, five times weekly and after 3 h, they received CCl4 at the same level given in second batch. The experimental animals of fourth batch were fed only with cinnamon, at the same dose given in third batch. Frogs of fifth batch were fed with 0.1 mL of corn oil, at the same dose administrated in second batch.
Haematological and biochemical measurements: After 2, 4 and 6 weeks, blood specimens were withdrawn from the experimental animals by cardiac puncture. Specimens of blood were collected into heparinized capillary tubes and non-heparinized tubes. The blood in the heparininzed capillary tubes were immediately used for haemtological measurements. Heamatological parameters analyzed were red blood cell count (RBC), haemoglobin concentration (Hb), haematocrit value (Hct), mean cell volume (MCV), mean corpuscular haemoglobin (MCH) and mean corpuscular haemoglobin concentration (MCHC). Red blood cell count was estimated according to the classical method using the Neubauer haemocytometer. Haemoglobin concentration was measured by Blood Haemoglobin Photometer System (HemoCue AB, Angelholm, Sweden). Haematocrit value was estimated using a microcapillary reader. The MCV, MCH and MCHC values were calculated by standard equations (Platt, 1969). The blood in non-heparinized tubes was centrifuged at 2000 rpm for 15 min. Sera were then stored at 4°C prior to determination of glutamic oxaloacetic acid transaminase (GOT) and glutamic pyruvic acid transaminase (GPT). Serum used in each observation is collected from 4 samples. GPT and GOT were analyzed by using BM/Hitachi system 717 Automatic Analyzer.
Hepatosomatic index: Livers from each batch were excised and weighed for the determination of hepatosomatic index (HSI) at 2, 4 and 6 weeks. The HIS was calculated by the following equation:
Statistical analysis: All the grouped data were statistically evaluated using the Statistical Package for Social Sciences (SPSS) version 12.0. Hypothesis methods one way analysis of variance (ANOVA) followed by least significant test. p-values of ≤0.05 were considered to indicate statistical significance. All results were expressed as mean±standard deviation (SD) for five observations in each batch.
RESULTS
The influences of oral administration of CCl4, cinnamon plus CCl4, cinnamon and corn oil on haematological, biochemical and hepatosomatic index values of the frog, Rana ridibunda, were represented in Fig. 1-9.
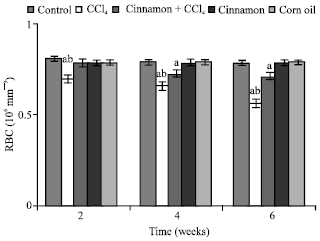 | |
| Fig. 1: | Number of red blood cells (RBC), n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
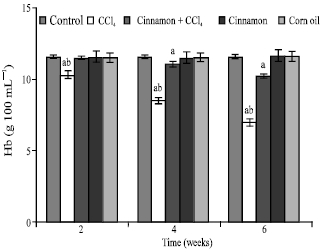 | |
| Fig. 2: | Heamoglobin (Hb) concentrations, n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
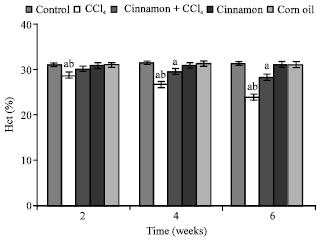 | |
| Fig. 3: | Haematocrit (Hct) levels, n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
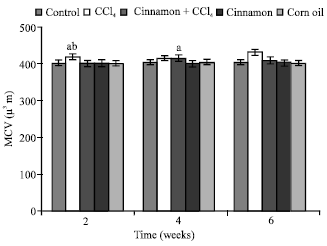 | |
| Fig. 4: | Mean cell volume (MCV) values, n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
In comparison with control and cinnamon plus CCl4 batches, significant decreases of RBC, Hb, Hct, MCHC and increases of GPT values were observed in CCl4-exposed batch at all experimental periods. Also, GOT and HIS levels were significantly elevated, while MCH values were decreased at second and last periods. MCV values were only increased at first period.
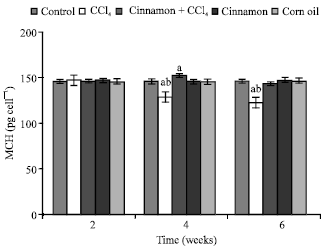 | |
| Fig. 5: | Levels of mean corpuscular haemoglobin (MCH), n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
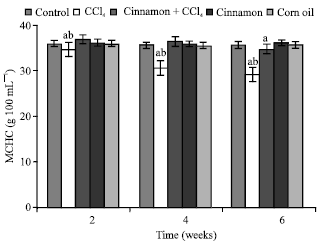 | |
| Fig. 6: | Mean corpuscular haemoglobin concentration (MCHC) values, n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
In comparison with control batch, significant decreases of RBC, Hb Hct and increases of GPT, GOT and HIS values were observed in frogs treated with cinnamon plus CCl4 at 2 and 6 weeks. MCV and MCH values were statistically elevated at second period.
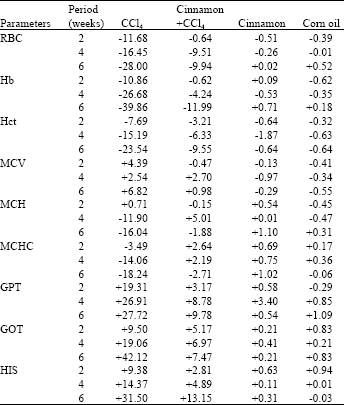
| Table 1: | Percentage changes of haematological, biochemical and hepatosomatic index values of the frog, Rana ridibunda treated with CCl4, cinnamon extract plus CCl4, cinnamon extract and corn oil for 2,4 and 6 weeks, from corresponding control data |
 | |
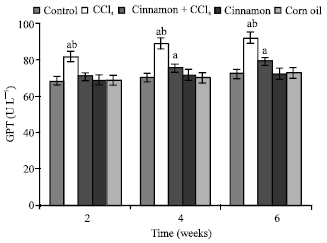 | |
| Fig. 7: | Levels of blood gultamic pyruvic acid transaminase (GPT), n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
MCHC values were declined at last period. No statistically significant differences were noted in these parameters in all animals treated with cinnamon extract (batch 4) or corn oil (batch 5).
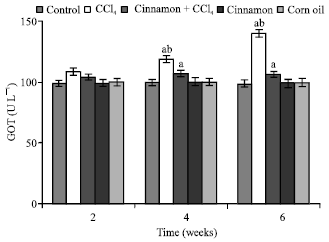 | |
| Fig. 8: | Blood gultamic oxaloacetic acid transaminase (GOT) values, n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
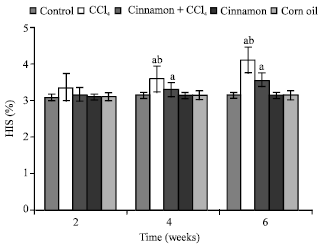 | |
| Fig. 9: | Hepatosomatic index (HIS) levels, n = 5, in the frog, Rana ridibunda, treated with CCl4, cinnamon plus CCl4, cinnamon and corn oil for 2, 4 and 6 weeks. Significance levels (ap≤0.05; one way ANOVA) shown for difference between control and treated batches. Significance levels (bp≤ 0.05; one way ANOVA) shown for difference between batches exposed to CCl4 and cinnamon plus CCl4 |
From Table 1, the percentage changes (increase or decrease) were more pronounced in the batch exposed to CCl4 only. Additionally, these changes in cinnamon plus CCl4 batch tended to be lower than CCl4 treated animals (batch 2).
DISCUSSION
The present study was conducted to evaluate the beneficial effects of cinnamon extract on physiological disturbances induced by CCl4 exposure for 2, 4 and 6 weeks. CCl4 exposure induced anemia is characterized by severe reduction in RBC count at all experimental periods. In addition, the observed decline of Hb, HCT, MCH and MCHC is similarly consistent with previous reports of anaemia in CCl4-exposed experimental animals (Mortiz and Pankow, 1989; Fujita, 1990; Mandal et al., 1998; Marel and Kanter, 2003). Moreover, blood is the first tissue to be affected by CCl4, as a result several deleterious effects on the haematological and biochemical parameters have been reported from different investigations (Farombi et al., 1997; MacDonald-Wicks and Garg, 2002; Guo et al., 2000; Satturwar et al., 2003; Wu et al., 2004; Chidambara Murthy et al., 2005). The present data provide evidence for the adverse influence of CCl4 on the liver functions when GOT and GPT were elevated in the serum. Since the determination of these enzymes was a good indicator for pathological manifestation of liver dysfunction, there was no doubt that CCl4 induces liver damage in the experimental animals. In addition, Several studies reported that blood GOT and GPT values were significantly elevated in CCl4-treated sheep (Alemu et al., 1977; Al-Waili, 2003), fishes (Casillas et al., 1983; Krajnovic-Ozretic and Ozretic, 1987; Folmar et al., 1993), rabbits (Zavodink et al., 1991; Kapuscinski et al., 1993), mice (Datta et al., 1998; Hewawasam et al., 2003; Liu et al., 2003; Das et al., 2004; Moreno and Muriel, 2006), rats (Sur-Altiner and Yenic, 2000; Kang et al., 2001; Sotelo-Felix et al., 2002; Valcheva-Kuzmanova et al., 2004; Kim et al., 2005; Baheti et al., 2006; Nagao et al., 2006) and guinea pigs (Sato et al., 2004). Liver, being the primary site for biotransformation of foreign compounds, is particularly vulnerable to these chemical assaults. Various enzymes are prone to the actions of intoxicants and their metabolites. In most of the cases, these enzymes leak from necrotic hepatocytes into the blood stream in abnormal amounts. Several of these soluble enzymes have been considered as indicators of liver function and damage (Kulkarni and Hodgson, 1980). The effects of CCl4 on hepatocytes, depending on dose and exposure time, are manifested histologically as hepatic steatosis (e.g., fatty infiltration), centrilobular necrosis and ultimately, cirrhosis. Hepatic steatosis of the liver is a multifactorial phenomenon thought to be caused by a blockage of lipoprotein secretion (Recknagel et al., 1960; Junnila et al., 2000), impaired synthesis or peroxidation of phospholipids, or both (Shimizu, 1969; Terao et al., 1984; Farombi, 2000), the toxic effects of free alkyl radicals on cell membranes (Tomasi et al., 1980; James et al., 1986) and disturbances in methylation reactions (Varela-Moreiras et al., 1995). The endoplasmic reticulum and mitochondria have been shown to be involved in cell damage (Oberling and Rouiller, 1956; Recknagel and Malamed, 1958; Afifi and MacMillan, 1992; Dai et al., 2001; Bhadauria et al., 2002; Che et al., 2004). The metabolic effects of CCl4 inside mitochondria have been described and it has been found that damage to the calcium pump in mitochondria (Brattin et al., 1984; Albano et al., 1985; Tang et al., 2005, 2006) is dependent upon haloalkylation. However, the profound accumulation of fat following CCl4 poisoning is considered independent of mitochondrial damage (Artizzu and Dianzani, 1962; Reynolds et al., 1962; Cunnane, 1987). The fatty infiltration of the liver is thought to develop as a result of the action of free alkyl radicals on biomembranes that in turn cause haloalkylation-dependent blocking at the exit of the lipoprotein micelles from the Golgi apparatus (Poli et al., 1987). Additionally, CCl4 has been widely used for inducing experimental hepatic damage due to free radical formation during its metabolism by hepatic microsome, which is turn to causes the lipid peroxidation of cellular membrane leading to the necrosis of hepatocytes (Sherlock, 1981). Wu and Norton (1996) demonstrated that liver microsomal oxidizing systems connected with cytochrome P-450 produce reactive metabolites of CCl4 such as trichloromethyl radical (CCl3) or trichloroperoxyl radical (CCl3O3). These radicals cause lipid peroxidation which produces hepatocellular damage and enhanced production of fibrotic tissue. This is evident from the elevated activities of the serum marker enzymes such as GOT and GPT. The observation of an elevated of HIS values in CCl4- treated frogs can also be attributed to liver damage. Several authors reported that the treatment with CCl4 caused significant increases in the liver weight (Kataria and Singh, 1997; Perez-Trueba et al., 2003; Raju et al., 2003; Nagao et al., 2006; Y et al., 2006).
The present investigation shows that the extract of cinnamon has a lowering effect on the studied parameters in CCl4-treated frogs, but the exact mechanism by which this action is elicited is not known. The published literature reveals different approaches for preventing the harmful effects of CCl4 to liver using various herbal extracts (Sur-Altiner and Yenic, 2000; Bhadauria et al., 2002; Hewawasam et al., 2003; Liu et al., 2003; Baheti et al., 2006; Tang et al., 2006). Cinnamon's unique healing abilities come from three basic types of components in the essential oils found in its bark. These oils contain active components called cinnamaldehyde, cinnamyl acetate and cinnamyl alcohol, plus a wide range of other volatile substances. Moreover, cinnamon constituents posses antioxidant action and may prove against free radical damage to cell membrane (Lee and Shibamoto, 2002; Dragland et al., 2003; Jayaprakasha et al., 2003). In addition, the present results suggest that cinnamon has the protective role against hepatotoxicity induced by CCl4 in the frog and the antioxidative influences of cinnamon components may play an important role in cell protection from enhancement of per-oxidative injuries.
In summary, the present findings reveal that the active roles of cinnamon suppress the physiological alterations is an interesting and promising topic for future study. It may therefore be suggested from the evidences from the present investigation, that cinnamon supplementation may give some beneficial results for people with some liver diseases. Finally, further investigation is needed to find out the exact mechanism of cinnamon reaction with CCl4 and its metabolites.
REFERENCES
- Al-Waili, N.S., 2003. Intravenous and intrapulmonary administration of honey solution to healthy sheep: Effects on blood sugar, renal and liver function tests, bone marrow function, lipid profile and carbon tetrachloride-induced liver injury. J. Med. Food, 6: 231-247.
CrossRefDirect Link - Bhadauria, M., A. Jadon, A. Sharma and S. Shukla, 2002. Effect of propriety herbal formulation against chronic carbon tetrachloride induced hepatotoxicity. Indian J. Exp. Biol., 40: 1254-1259.
Direct Link - Che, Y., T. Xu, M. Jiang and Y. Jia, 2004. Protective effect of ganfukang capsule on hepatocytes in rats with experimental hepatic fibrosis. Zhang. Yao. Cai, 27: 428-429.
Direct Link - Chidambara Murthy, K.N., J. Rajesha, A. Vanitha, M.M. Swamy and G.A. Ravishankar, 2005. Protective effect of Dunaliella salina. A marine micro alga, against carbon tetrachloride-induced hepatotoxicity in rats. Hepatol. Res., 33: 313-319.
PubMedDirect Link - Dai, W., S. Sato and G. Asano, 2001. The protective effect of hepatocyte growth-promoting factor (pHGF) against carbon tetrachloride-induced acute liver injury in rats. II. Protective effects on cell membrane injury. J. Nippon. Med. Sch., 68: 154-164.
Direct Link - Das, R.K., S. Das and S. Bhattacharya, 2004. Protective effect of diphenylmethyl selenocyanate against carbon tetrachloride-induced hepatotoxicity in vivo. J. Environ. Pathol. Toxicol. Oncol., 23: 287-296.
Direct Link - Datta, S., K. Basu, S. Sinha and P. Bhattacharyya, 1998. Hepatoprotective effect of a protein isolated from Cajanus indicus (Spreng) on carbon tetrachloride induced hepatotoxicity in mice. Indian J. Exp. Biol., 36: 175-181.
Direct Link - Dogukan, A., N. Akpolat, H. Celiker, N. Ilhan, I.H. Bahcecioglu and A.I. Gunal, 2003. Protective effect of interferon-alpha on carbon tetrachloride-induced nephrotoxicity. J. Nephrol., 16: 81-84.
Direct Link - Dragland, S., H. Senoo, K. Wake, K. Holte and R. Blomhoff, 2003. Several culinary and medicinal herbs are important sources of dietary antioxidants. J. Nutr., 133: 1286-1290.
CrossRefPubMedDirect Link - Farombi, E.O., J.O. Nwankwo and G.O. Emerole, 1997. Possible modulatory effect of browned yam flour diet on chemically-induced toxicity in the rat. Food Chem. Toxicol., 35: 975-979.
Direct Link - Farombi, E.O., 2000. Mechanisms for the hepatoprotective action of kolaviron: Studies on hepatic enzymes, microsomal lipids and lipid peroxidation in carbontetrachloride-treated rats. Pharmacol. Res., 42: 75-80.
Direct Link - Fujita, K., 1990. A study on erythrocyte-membrane osmotic resistance and periodontal changes in rats treated with carbon tetrachloride. Shikwa. Gakuh., 90: 727-743.
Direct Link - Guo, T.L., J.A. McCay, R.D. Brown, D.L. Musgrove and D.R. Germolec et al., 2000. Carbon tetrachloride is immunosuppressive and decreases host resistance to Listeria monocytogenes and Streptococcus pneumoniae in female B6C3F1 mice. Toxicology, 154: 85-101.
CrossRef - Ha, K.T., S.J. Yoon, D.Y. Choi, D.W. Kim, J.K. Kim and C.H. Kim, 2005. Protective effect of Lycium chinense fruit on carbon tetrachloride-induced hepatotoxicity. J. Ethnopharmacol., 96: 529-535.
Direct Link - Hewawasam, R.P., K.A. Jayatilaka, C. Pathirana and L.K. Mudduwa, 2003. Protective effect of Asteracantha longifolia extract in mouse liver injury induced by carbon tetrachloride and paracetamol. J. Pharm. Pharmacol., 55: 1413-1418.
CrossRefDirect Link - Hui, A.Y., W.K. Leung, H.L.Y. Henry Chan and F.K.L. Chan et al., 2006. Effect of celecoxib on experimental liver fibrosis in rat. Liver Intl., 26: 125-136.
Direct Link - Jayaprakasha, G.K., L. Jagan Mohan Rao and K.K. Sakariah, 2003. Volatile constituents from Cinnamomum zeylancium fruit stalks and their antioxidant activities. J. Agric. Food Chem., 51: 4344-4348.
Direct Link - Jiko, M., I. Yano, M. Okuda and K. Inui, 2005. Altered pharmacokinetics of paclitaxel in experimental hepatic or renal failure. Pharm. Res., 22: 228-234.
Direct Link - Junnila, M., T. Rahko, A. Sukura and L.A. Lindberg, 2000. Reduction of carbon tetrachloride-induced hepatotoxic effects by oral administration of betaine in male Han-Wistar rats: A morphometric histological study. Vet. Pathol., 37: 231-238.
CrossRefDirect Link - Kahkonen, M.P., A.I. Hopia, H.J. Vuorela, J.P. Rauha, K. Pihlaja, T.S. Kujala and M. Heinonen, 1999. Antioxidant activity of plant extracts containing phenolic compounds. J. Agric. Food Chem., 47: 3954-3962.
CrossRefPubMedDirect Link - Kang, J.O., S.J. Kim and H. Kim, 2001. Effect of astaxanthin on the hepatotoxicity, lipid peroxidation and antioxidative enzymes in the liver of CCl4-treated rats. Methods Find Exp. Clin. Pharmacol., 23: 79-84.
Direct Link - Kapuscinski, J., J. Kuroszczyk, J.M. Liniecki, M. Bienkiewics, K. Zielinski and K. Tuszyner, 1993. Experimental toxic liver damage and hepatic plasma clearance of 99mTc-mebrofenin (iminodiacetate derivative). I. early, acute CCl4-induced liver damage in rabbits. Pol. J. Occup. Med. Environ. Health, 6: 169-183.
Direct Link - Kataria, M. and L.N. Singh, 1997. Hepatoprotective effect of Liv-52 and kumaryasava on carbon tetrachloride induced hepatic damage in rats. Indian J. Exp. Biol., 35: 655-657.
Direct Link - Kim, N.Y., M.K. Lee, M.J. Park, S.J. Kim and H.J. Park et al., 2005. Momordin Ic and oleanolic acid from Kochiae Fructus reduce carbon tetrachloride-induced hepatotoxicity in rats. J. Med. Food, 8: 177-183.
CrossRefPubMedDirect Link - Kim, S.H., S.H. Hyun and S.Y. Choung, 2006. Anti-diabetic effect of cinnamon extract on blood glucose in db/db mice. J. Ethnopharmacol., 104: 119-123.
CrossRefDirect Link - Larson, R.A., 1988. The antioxidants of higher plants. Phytochemistry, 27: 969-978.
CrossRefDirect Link - Lee, K.G. and T. Shibamoto, 2002. Determination of antioxidant potential of volatile extracts isolated from various herbs and spices. J. Agric. Food Chem., 50: 4947-4952.
CrossRefPubMedDirect Link - Liu, Y.G., H.C. Chen and Y.P. Jiang, 2003. Protective effect of curcumin on experimental liver injury in mice. Zhongguo. Zhong. Yao. Za. Zhi., 28: 756-758.
Direct Link - MacDonald-Wicks, L.K. and M.L. Garg, 2002. Modulation of carbon tetrachloride-induced oxidative stress by dietary fat in rats. J. Nutr. Bioch., 13: 87-95.
Direct Link - MacDonald-Wicks, L.K. and M.L. Garg, 2002. Modulation of carbon tetrachloride-induced oxidative stress by dietary fat in rats. J. Nutr. Bioch., 13: 87-95.
Direct Link - Meral, I. and M. Kanter, 2003. Effects of Nigella sativa L. and Urtica dioica L. on selected mineral status and hematological values in CCl4- treated rats. Biol. Trace Element Res., 96: 263-270.
CrossRefPubMedDirect Link - Moreno, M.G. and P. Muriel, 2006. Inducible nitric oxide synthase is not essential for the development of fibrosis and liver damage induced by CCl4 in mice. J. Applied Toxicol., 26: 326-332.
Direct Link - Mortiz, R.P. and D. Pankow, 1989. [Effect of carbon tetrachloride and chloroform on hematologic parameters in rats]. Folia Haematologica, 116: 283-287, (In German).
PubMedDirect Link - Ozturk, F., M. Ucar, I.C. Ozturk, N. Vardi and K. Batcioglu, 2003. Carbon tetrachloride-induced nephrotoxicity and protective effect of betaine in Sprague-Dawley rats. Urology, 62: 353-356.
Direct Link - Perez-Trueba, G., C. Ramos-Guanche, B. Martinez-Sanches, I. Marquez-Hernandez, A. Giuliani and G. Martinez-Sanches, 2003. Protective effect of gossypitrin on carbon tetrachloride-induced in vivo hepatotoxicity. Redox. Rep., 8: 215-221.
Direct Link - Poli, G., E. Albano and M.U. Dianzani, 1987. The role of lipid peroxidation in liver damage. Chem. Phys. Lipids, 45: 117-142.
CrossRefDirect Link - Raju, K., G. Anbuganapathi, V. Gokulakrishnan, B. Rajkapoor, B. Jayakar and S. Manian, 2003. Effect of dried fruits of Solanum nigrum Linn against CCl4-induced hepatic damage in rats. Biol. Pharmaceut. Bull., 26: 1618-1619.
CrossRefPubMedDirect Link - Sato, N., S. Abe, T. Yamada, K. Iwasaki, Y. Ohtake and Y. Ohkubo, 2004. Relationship between liver injury and transglutaminase activities in guinea pigs and rats. Biol. Pharm. Bull., 27: 236-238.
Direct Link - Satturwar, P.M., S.V. Fulzele, S.B. Joshi and A.K. Dorle, 2003. Hepatoprotective activity of Haridradi ghrita on carbon tetrachloride-induced liver damage in rats. Indian J. Exp. Biol., 41: 1447-1451.
Direct Link - Shailajan, S., C. Naresh, R.T. Sane and M. Sasikumar, 2005. Effect of Asteracantha longifolia Nees. against CCl4 induced liver dysfunction in rat. Indian J. Exp. Biol., 43: 68-75.
Direct Link - Sotelo-Felix, J.I., D. Martinez-Fong and P. Muriel De la Torre, 2002. Protective effect of carnosol on CCl4-induced acute liver damage in rats. Eur. J. Gastroenterol. Hepatol., 14: 1001-1006.
Direct Link - Sur-Altiner, D. and B. Yenic, 2000. Effect of black tea on lipid peroxidation in carbon tetrachloride treated male rats. Drug Metabol. Drug Interact., 16: 123-128.
PubMedDirect Link - Taira, Z., K. Yabe, Y. Hamaguchi, K. Hirayama, M. Kishimoto, S. Ishida and Y. Ueda, 2004. Effects of Sho-saiko-to extract and its components, baicalin, baicalein, glycyrrhizin and glycyrrhetic acid, on pharmacokinetic behavior of salicylamide in carbon tetrachloride intoxicated rats. Food Chem. Toxicol., 425: 803-807.
CrossRef - Tang, X.H., J. Gao, F. Fang, J. Chen, L.Z. Xu, X.N. Zhao and Q. Xu, 2005. Hepatoprotection of oleanolic acid is related to its inhibition on mitochondrial permeability transition. Am. J. Chin. Med., 33: 627-637.
Direct Link - Tang, X.H., J. Gao, Y. Wang, Y.M. Fan and L.Z. Xu et al., 2006. Effective protection of Terminalia catappa L. leaves from damage induced by carbon tetrachloride in liver mitochondria. J. Nutr. Biochem., 17: 177-182.
CrossRefDirect Link - Valcheva-Kuzmanova, S., P. Borisova, B. Galunska, I. Krasnaliev and A. Belcheva, 2004. Hepatoprotective effect of the natural fruit juice from Aronia melanocarpa on carbon tetrachloride-induced acute liver damage in rats. Exp. Toxicol. Pathol., 56: 195-201.
CrossRef - Varela-Moreiras, G., E. Alonso-Aperte, M. Rubio, M. Gasso and R. Deulofeu et al., 1995. Carbon tetrachloride-induced hepatic injury is associated with global DNA hypomethylation and homocysteinemia: Effect of S-adenosylmethionine treatment. Hepatology, 22: 1310-1315.
CrossRefDirect Link - Velioglu, Y.S., G. Mazza, L. Gao and B.D. Oomah, 1998. Antioxidant activity and total phenolics in selected fruits, vegetables and grain products. J. Agric. Food Chem., 46: 4113-4117.
CrossRefDirect Link - Weisburger, E.K., 1977. Carcinogenicity studies on halogenated hydrocarbons. Environ. Health Perspect., 21: 7-16.
Direct Link - Wilson, J.T., 1954. Influence on the offspring of altered physiologic states during pregnancy in the rat. Ann. N.Y. Acad. Sci., 57: 517-525.
CrossRefDirect Link - Wu, Y.W., K.D. Chen and W.C. Lin, 2004. Effect of Ganoderma tsugae on chronically carbon tetrachloride-intoxicated rats. Am. J. Chin. Med., 32: 841-850.
Direct Link - Jyothi, Y., J.V. Kamath and M. Asad, 2006. Effect of hexane extract of Boswellia serrata oleo-gum resin on chemically induced liver damage. Pak. J. Pharmaceut. Sci., 19: 129-133.
Direct Link - Zavodnik, L.B., M.I. Bushma, P.I. Lukienko, G.Z. Abakumov, I.V. Zverinskii and V.M. Tsyrkunov, 1991. Diethylnicotinamid (cordiamine) stabilization of the liver hydroxylating function in rabbits with CCl4 poisoning. Farmakol. Toksikol., 54: 69-71.
Direct Link








