Research Article
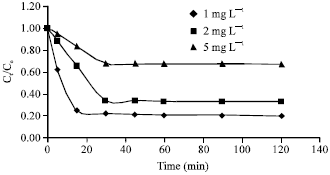
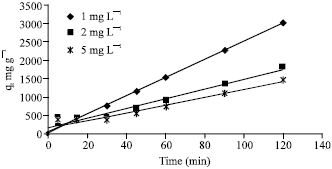
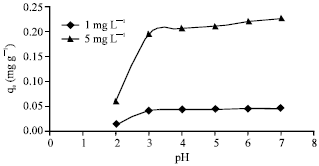
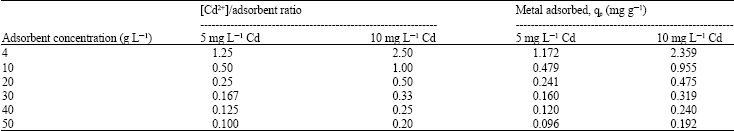
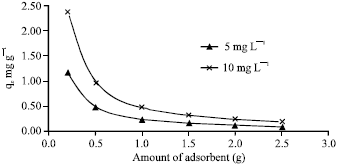
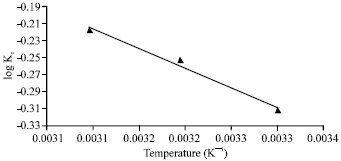
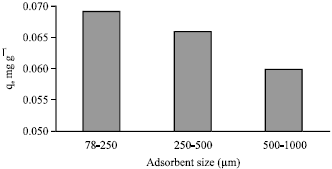
Adsorption of Cd(II) Ions from Aqueous Solutions by Lalang (Imperata cylindrica) Leaf Powder: Effect of Physicochemical Environment
Faculty of Applied Sciences, Universiti Teknologi MARA, 26400, Jengka, Pahang, Malaysia
M.Z.A. Yahya
Faculty of Applied Sciences, Universiti Teknologi MARA, 40450, Shah Alam, Selangor, Malaysia
H. Zakaria
Faculty of Applied Sciences, Universiti Teknologi MARA, 26400, Jengka, Pahang, Malaysia
S.C. Ibrahim
Faculty of Applied Sciences, Universiti Teknologi MARA, 40450, Shah Alam, Selangor, Malaysia