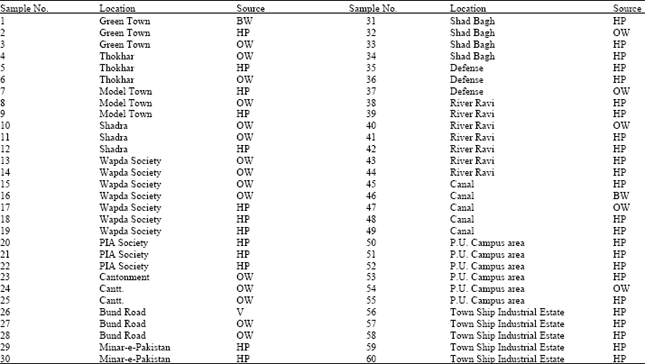
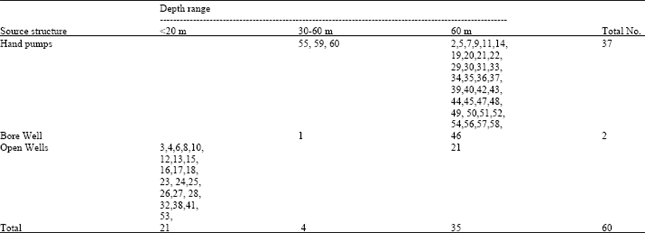
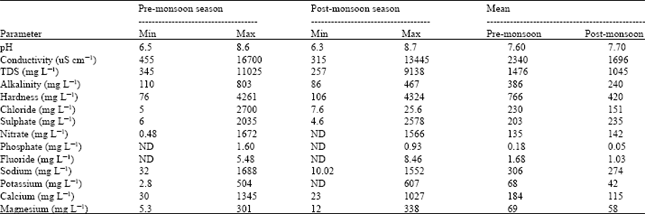
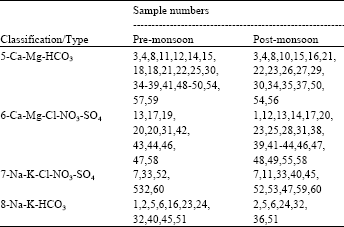
Research Article
Environmental Assessment of Ground Water Quality of Lahore Area, Punjab, Pakistan
Applied Chemistry Research Center, PCSIR, Labs, Complex, Lahore, Pakistan
Khalida Khan
Center for Integrated Mountain Research (CIMR), University of the Punjab, Lahore, Pakistan
Salma Rehman
Applied Chemistry Research Center, PCSIR, Labs, Complex, Lahore, Pakistan
Javad Iqbal
Institute of Chemistry, University of the Punjab, Lahore, Pakistan