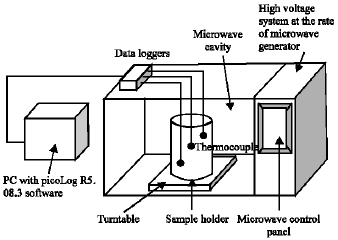
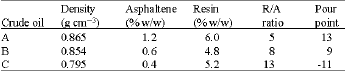
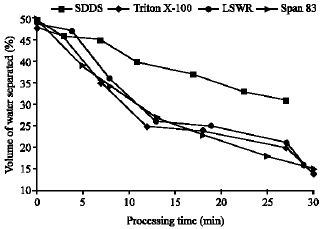
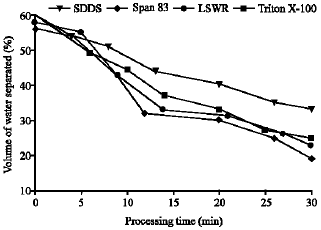
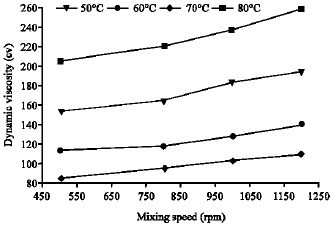
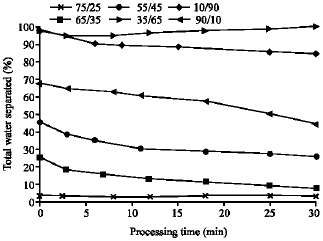
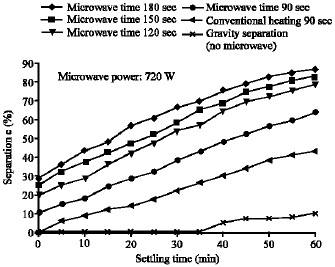
Research Article
Water-in-Crude Oil Emulsions: Its Stabilization and Demulsification
Faculty of Chemical and Natural Resources Engineering, Universitiy Malaysia Pahang, P.O. Box 12, UMP 25000, Kuantan, Pahang, Malaysia
R. Mohd. Yunus
Faculty of Chemical and Natural Resources Engineering, Universitiy Malaysia Pahang, P.O. Box 12, UMP 25000, Kuantan, Pahang, Malaysia
H. Anwaruddin
Faculty of Chemical and Natural Resources Engineering, Universiti Malaysia Pahang, P.O. Box 12, UMP 25000, Kuantan, Pahang, Malaysia