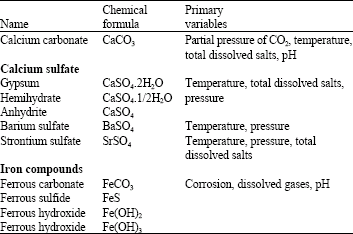
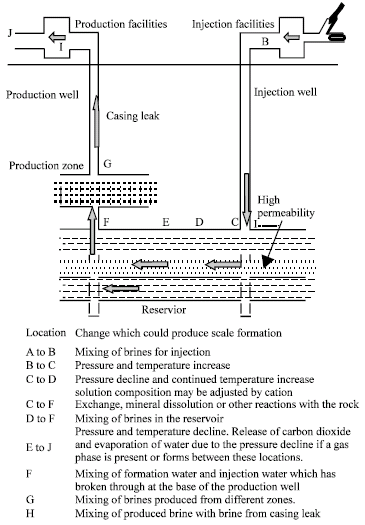
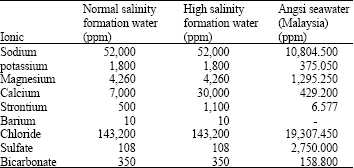
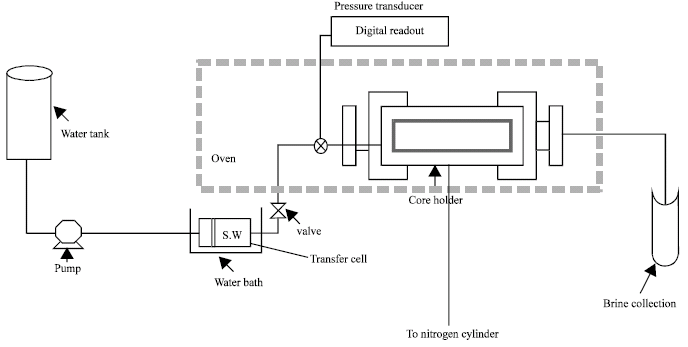
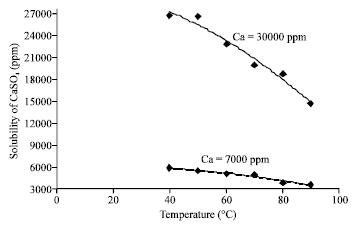
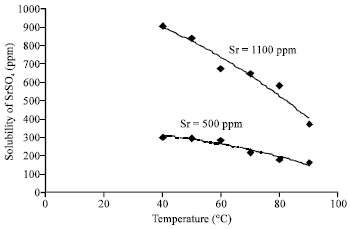
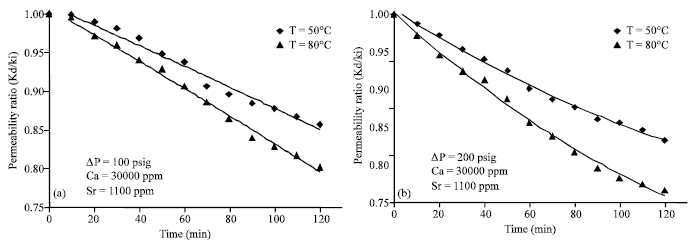
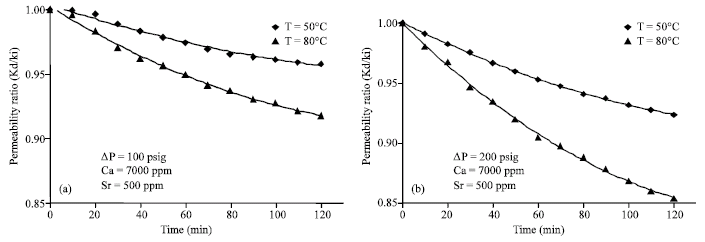
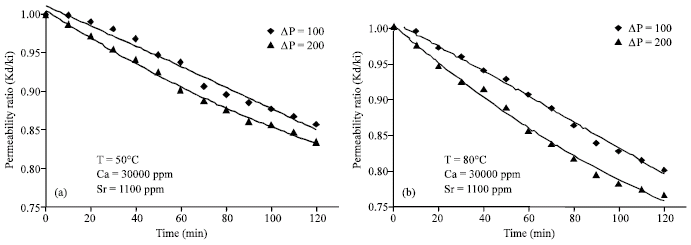
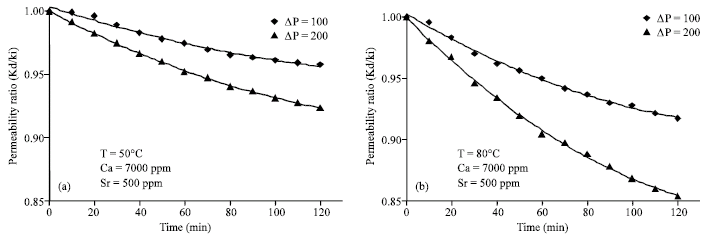
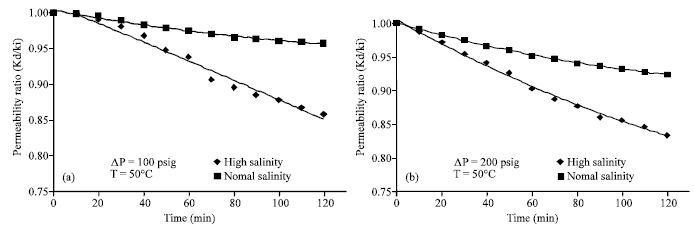
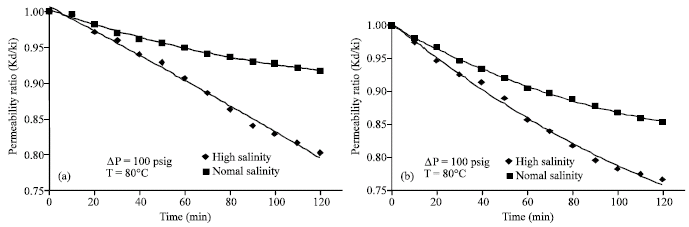
Research Article
Scale Formation in Oil Reservoir During Water Injection at High-Salinity Formation Water
Faculty of Chemical and Natural Resources Engineering,Universiti Teknologi Malaysia, 81310 Skudai, Johor, Malaysia
Abu Azam Mohd Yassin
Faculty of Chemical and Natural Resources Engineering,Universiti Teknologi Malaysia, 81310 Skudai, Johor, Malaysia