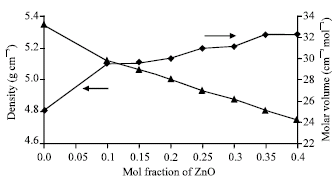
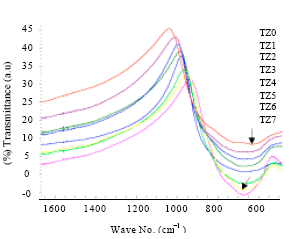
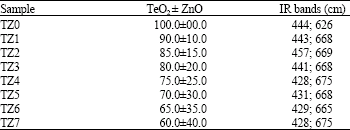
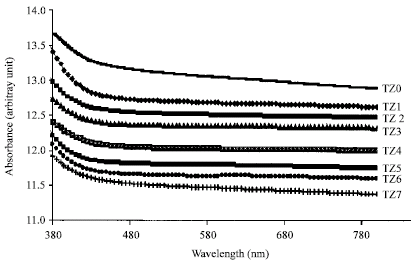
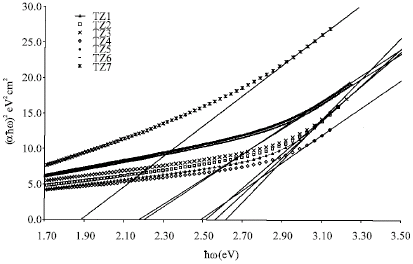
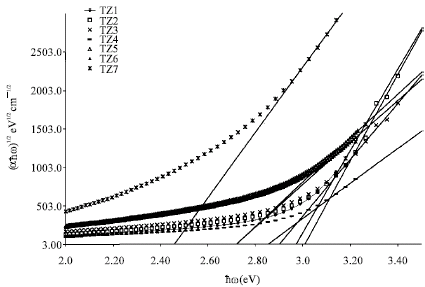
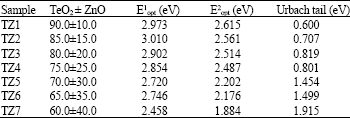
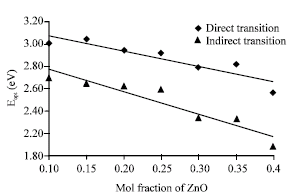
Research Article
IR and UV Spectral Studies of Zinc Tellurite Glasses
Laboratory of Ultrasonics and Glass, Department of Physics, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
H.A.A. Sidek
Laboratory of Ultrasonics and Glass, Department of Physics, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
A.T. Zainal
Laboratory of Ultrasonics and Glass, Department of Physics, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
H. Mohd Zobir
Department of Chemistry, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia