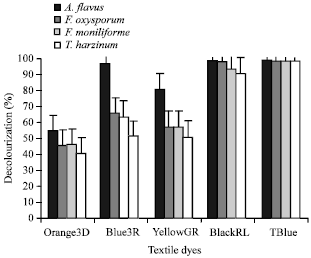
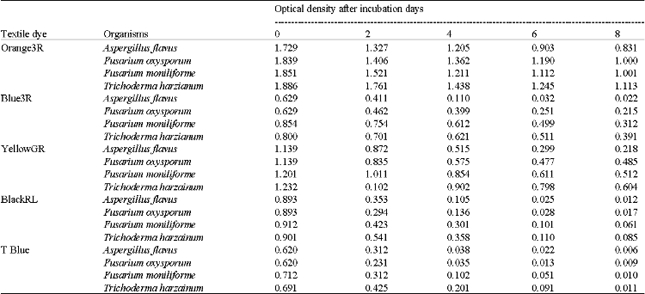
Case Study
Isolation and Evaluation of Indigenous Soil Fungi for Decolourization of Textile Dyes
Department of Studies in Environmental Science Manasagangotri, University of Mysore Karnataka, India 570006
G.V. Venkataramana
Department of Studies in Environmental Science Manasagangotri, University of Mysore Karnataka, India 570006
S.T. Girish
Department of Studies in Botany and Microbiology, Yuvarajas College University of Mysore, India
V.B. Raghavendra
Department of Studies in Applied Botany and Biotechnology, Manasagangotri, University of Mysore, India
P. Shivashankar
Department of Studies in Environmental Science Manasagangotri, University of Mysore Karnataka, India 570006