Research Article
Physics Properties Comparison Between Undoped ZnO and AZO, IZO Doped Thin Films Prepared By Spray Pyrolysis
Department of Physics, Electron Microscope Laboratory, USTOMB University, BPI 505 Oran, Algeria
A. Sanchez-Juarez
Centro de Investigacion en Energias-UNAlVT, Temixco, Morelos 625 80, Mexico
M.S. Aida
Department of Physics, Thin Films and Plasma Laboratory, Mentouri University, 25000 Constantine, Algeria









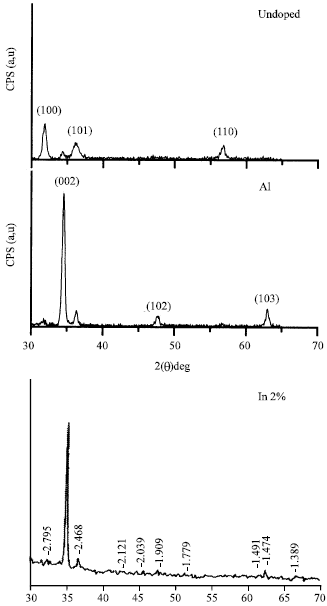
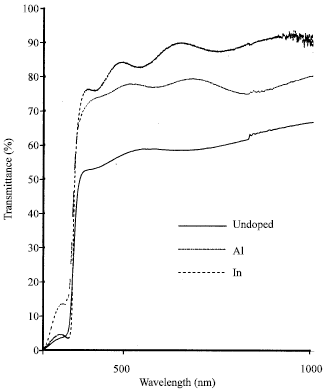
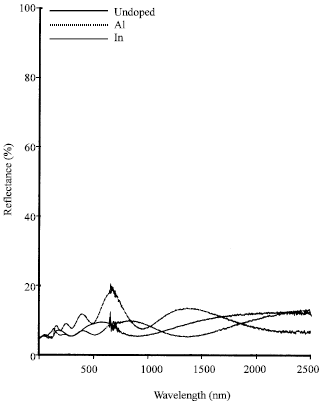
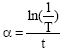
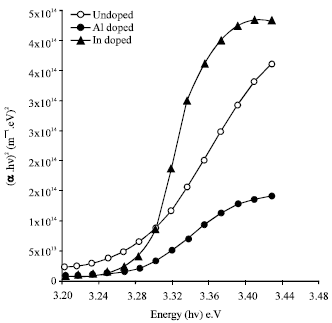
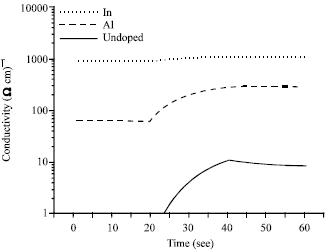
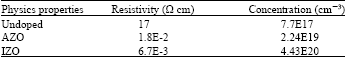

Shadia Jamil Ikhmayies Reply
Please I want to reuse Fig.1, Fig.2, and Fig.4 in a conference presentation, then in a journal paper. How I can get permissions to reprint them?
Kind regards