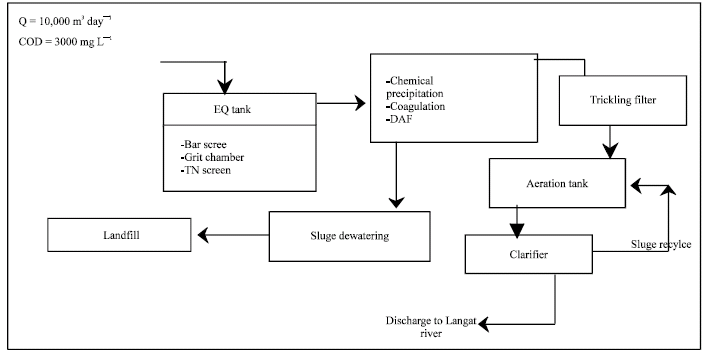
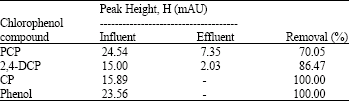
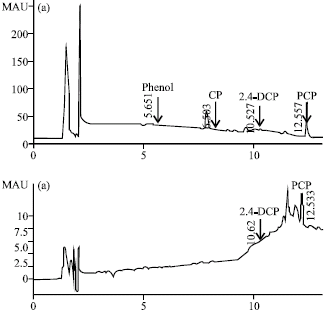
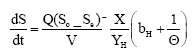Research Article
Chloro-Organics in Papermill Effluent: Identification and Removal by Sequencing Batch Biofilm Reactor
Department of Chemical and Process Engineering, Universiti Rebangsaan Malaysia, 43600 UKM Bangi, Malaysia
A.Y. Zahrim
Chemical Engineering Programme, School of Engineermg and Information Technology, Universiti Malaysia Sabah, Locked Bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
Azizah Abu Bakar
Department of Chemical and Process Engineering, Universiti Rebangsaan Malaysia, 43600 UKM Bangi, Malaysia