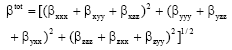Research Article
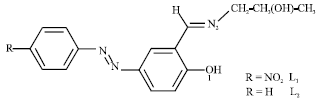
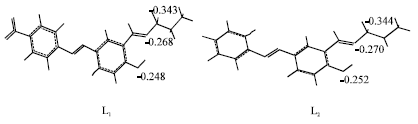
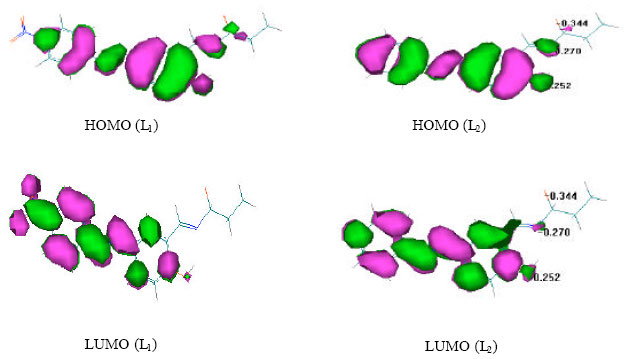
Some Quantum Chemical Study about the Second-Order Non-linearity of Two Imino Chromophores Containing Salen Group
Department of Chemistry, Shahid Bahonar University of Kerman, Iran
Samira Saeid-Nia
Department of Chemistry, Shahid Bahonar University of Kerman, Iran
Niaz Monadi
Department of Chemistry, Shahid Bahonar University of Kerman, Iran
Hojatollah Khabazzadeh
Department of Chemistry, Shahid Bahonar University of Kerman, Iran