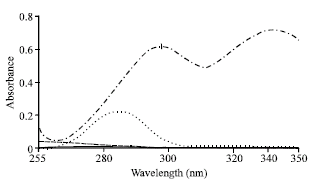
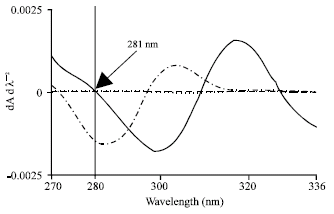
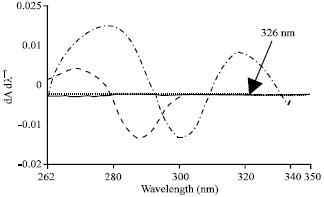
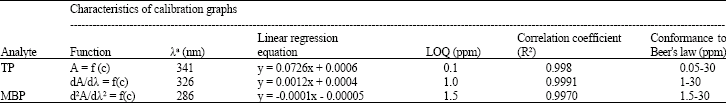Research Article
UV Spectrometric Study on a Synergetic System of Polymer Additives
Department of Analytical Chemistry, Faculty of Chemistry, University of Bucharest, 4-12 Regina Elisabeta Blvd, 030018, Sect. 3-Bucharest, Romania
Laura Alexandrescu
Research Development National Institute for Textile and Leather Division Leather and Footwear Research Institute 93 Ion Minulescu St., Sect 3, 031215-Bucharest, Romania
Valentina Constantinescu
Department of Analytical Chemistry, Faculty of Chemistry, University of Bucharest, 4-12 Regina Elisabeta Blvd, 030018, Sect. 3-Bucharest, Romania