ABSTRACT
This study aims at assessing the effect of different treatments on Kochi tap water of Japan with different materials belonging to various parameters. Granular ceramics and alumina having electrochemical charges at the surface were produced in a fluidization system with tap water. Ceramics and alumina were used as fluidizing agents in the experiment and three different amounts of these materials for each experiment were passed through the fluidizing layer in three separate selected periods. Results showed that after each treatment with Kochi tap water of Japan, pH and surface tension (ST) increased, oxidation-reduction potential (ORP) decreased as compared with untreated water and the values of electrical conductivity (EC) did not change significantly in the same experimental conditions. During fluidization process of water with ceramics and alumina, water flows through the granular substances and every particle comes into contact with each other in a frictional way. Resultantly, the raising temperature of disperse medium and oxidation-reduction reactions have been taken place which were considered as the possible reasons for changing the values of pH, ORP, ST and EC in this experiment. After treatments, other than the changing values of ST and EC, the increasing and decreasing tendencies of pH and ORP from their initial values were preferable for drinking and agriculture.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2006.1822.1826
URL: https://scialert.net/abstract/?doi=jas.2006.1822.1826
INTRODUCTION
Water environment protection is becoming a growing concern as water environment has polluted quickly. Water is a complex and heterogeneous in nature. Generally, in the nature without adding anything, the chemical components in a particular location of a water body are more or less stable, alternatively some physical properties are always spontaneously changeable (Azad et al., 2004). Increasing population and their expanded living standards will cause ever-increasing demands for good quality of raw water (Bouwer, 1994). A judicial and integrated approach to water management that would ensure the best possible utilization of the waters available and maximize the benefits for all concerned (Ahmed, 2002). For the improvement of water quality innovative techniques should be find out without using chemicals. Granular ceramics and alumina were used in different treatments through a fluidization system with Kochi tap water of Japan to examine the characteristics of water in the study. Ishikawa et al. (1998; 2005), Tamura et al. (1999), Azad and Ishikawa (2003; 2004) worked with granular ceramics in a fluidization treatment system for the improvement of water quality. Alumina is another treatment material which used in this experiment to compare its activities with granular ceramics in treating water.
The water treatment design elements are the volume of fluidization layer, fluidization time, speed of fluidization and the amount of ceramics and alumina. The objectives of the experiment were to study the various properties of water and to compare the potentiality of ceramics and alumina in changing some characteristics of water to improve water quality through the fluidization treatment system without using any chemicals concerning with environmental protection.
MATERIALS AND METHODS
Granular ceramics and alumina were applied to treat Kochi tap water in a fluidization system. In the fluidization method different amount of these materials (ceramics and alumina) were kept inside the fluidization layer at different levels viz., apparently full volume (100%), half (50%) volume and one third (30%) level volume of the fluidization layer.
For the experiment, ceramics and alumina were weighed in a balance in three replications. Bulk density was calculated by using the following formula.
Here,
| ρ | = | Bulk density |
| m | = | Mass of the materials |
| v | = | Volume of the materials |
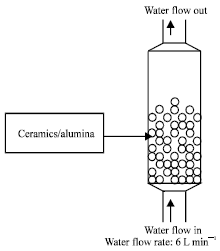 | |
| Fig. 1: | Water treatment cell |
| Table 1: | Different measured units of ceramics |
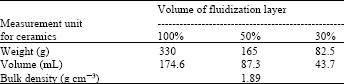 | |
| Table 2: | Different measured units of alumina |
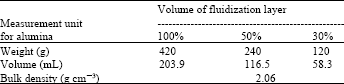 | |
Granular ceramics: Granular ceramics were made by using the powder of quartz porphyry and natural soils as a bonding agent, sintering at 1200°C (Ishikawa and Tamura, 1996 ). And these were 2 to 3 mm in diameter, round shaped with smooth surface. Moh`s hardness of granular ceramics is 6.8. For granular ceramics 100% volume of fluidization layer contained 330 g, 50% contained 165 g and 30% contained 82.5 g of ceramics. Weight, volume and bulk density of ceramics have been shown in the Table 1.
Alumina: The same size and diameter (2 mm) of granular alumina is heavier than that of ceramics. For being this reason, here 100, 50 and 30% fluidization layer of volume contained bigger amount of alumina as well as the volume and density i.e., 100% contained 420 g, 50% contained 240 g and 30% contained 120 g of alumina (Table 2). Granular alumina also round shaped with smooth surface.
Water treatment apparatus: A model of fluidization process was followed for the treatment of water in the study. This process is widely used in powder industry (Horio and Mori, 1999). Later, Ishikawa et al. (1996) and Azad et al. (2003; 2004) performed experiments following the theory of this model for treating water with this apparatus. The fluidizing cell of the apparatus for water treatment as shown in the Fig. 1 was constructed for experiments.
The upper and lower sides of a fluidization layer were partitioned with stainless steel nets. Into the partition, each material (granular ceramics and alumina) of indicated amounts were inserted. The apparatus was equipped with a magnetic pump that pushes the water flow from the lower to the upper side so that the materials could be fluidized. For the apparatus, a circulation system was employed, by which water passing through the fluidization layer was fed back into the water tank and the flow rate could be controlled at 6 L min-1 with a valve.
Experimental procedure (measurement of pH, ORP, ST and EC): Ceramics and alumina were weighed separately for the apparent volume of 100, 50 and 30% of the fluidization layer/pipe. After setting up of each material of granular substances in different amounts in different volumetric pipes, the magnetic pump was switched on. Three various treatment periods viz., 10 and 5 min and one pass water through (30 sec) the fluidization layer were selected for each treatment separately. The changes in the state of Kochi tap water were examined under a constant condition temperature at 18°C. Some properties of supplied water samples were examined using different treatment by specific apparatus. The values of pH, oxidation-reduction potential (ORP), surface tension (ST) and electrical conductivity (EC) were measured with pH meter (PHL-10, DKK), ORP meter, tensometer (ST-1, SHIMADZU) and EC meter, respectively. The preparation of samples and all other analyses for the experiment were conducted and carried out in the laboratory of Biological and Environmental systems in Kochi University, Japan from March 2002 to March 2003.
RESULTS AND DISCUSSION
Table 3 shows different parameters of distilled water and treated distilled water in different conditions. After treatments with 82.5 g ceramics (30%), pH, ST and EC increased while ORP decreased as compared to untreated (defined as control) distilled water in every cases (Table 3).
The same trends of results are also shown for pH, ST, EC and ORP in using 120 g alumina (30% and Table 3) with all treatment conditions. The amount of 82.5 g ceramics and 120 g alumina occupied the same volume of the fluidization layer.
Treatment of tap water with ceramics: For the treatment of tap water with 330 g of ceramics in the fluidization system, there have been used three different periods e.g., one pass water through ceramics (around 30 sec), 5 and 10 min.
| Table 3: | Some characteristics of distilled water in control, ceramics and alumina treatment (fluidized) in different conditions |
 | |
| Temperature: 18°C | |
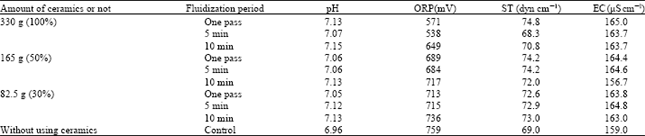
| Table 4: | Changing of different parameters of water in the fluidization treatment with different amount of ceramics in various treatment (fluidized) periods |
 | |
| Temperature: 18°C | |
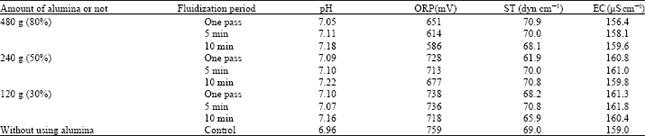
| Table 5: | Changing of different parameters of water in the fluidization treatment with different amounts of alumina in various treatment (fluidized) periods |
 | |
| Temperature: 18°C | |
Acidity (pH), ST, EC and ORP decreased in all fluidization conditions with ceramics compare to control. The values of pH and ST were 7.13 and 74.8 dyn cm-1 (Table 4) at the treatment of one pass water through ceramics and both these were decreased with the treatment of 5 min fluidization period, then increased at the end of 10 min fluidization treatment in both cases. ORP increased after each experimental period while decrement occurred in EC.
The values of pH, ST and EC were greater and ORP was smaller than control in using 165 g of ceramics (Table 4) in the fluidization treatment with Kochi tap water in the same constant temperature at 18°C. The values of each parameter did not change significantly with the treatment of one pass ceramics and 5 min fluidization period. Alternatively, significant changes of pH, ST and EC were occurred in using 10 min fluidization period. Acidity (pH) and ORP increased, ST decreased at this treatment condition than the other two.
In using 82.5 g ceramics for the treatment, all measured parameters except ORP were greater than the control (Table 4) in all the treatment conditions. Within these treatments the values of pH, ORP and ST were increased gradually after the treatments of one pass, 5 min and 10 min periods respectively. EC changed anomalously with a decreased mark at the end of 10 min fluidization treatment.
Acidity (pH 7.13), ST (74.8 dyn cm-1) and EC (165 μS cm-1) were greater and ORP (571 mV) smaller in using 330 g ceramics than by used 165 g (pH 7.06, ST 74.2 dyn cm-1, EC 164.4 μS cm-1) and 82.5 g (pH 7.05, ST 72.6 dyn cm-1, EC 163.8 μS cm-1) of ceramics for the treatment of one pass ceramics. With the treatment of 5 min fluidization period pH, ORP and EC for fluidizing 82.5 g of ceramics were higher than the others (Table 4). EC values followed the increasing order of 330 >82.5 >165 g and pH was not changed significantly using all amounts of ceramics separately for 10 min treatment period. ORP increased in the order of 82.5 >165 >330 g for 10 min fluidization period. In this treatment time EC value became lowest (156.3 μS cm-1) in using 165 g of ceramics. And there was no significant fluctuation in EC value in between using 330 and 82.5 g (Table 4) of ceramics.
Treatment of tap water with alumina: Near about 100% volume of the fluidization layer contained 480 g of alumina as treatment materials. While using this amount of alumina with tap water in the fluidization process, the values of pH, EC increased, ORP and ST decreased with the treatment of one pass, 5 min and 10 min period, respectively (Table 5).
In the case of using 240 g (50%) alumina, pH and ST increased, ORP decreased and EC changed anomalously, but after the treatment for 10 min fluidization period EC reached around the lowest level at 159.8 μS cm-1 (Table 5).
In fluidizing 120 g of alumina with tap water in the treatment of one pass, 5 and 10 min pH, ORP and EC changed (Table 5) in the same trend chronologically as shown above results.
In all conditions for alumina irrespective of its weight pH increased and ORP decreased significantly and the value of EC became lower with the fluidization treatment for 10 min than the others.
Comparison between ceramics and alumina treatments: Acidity (pH) changed but not in a unified way using ceramics in all treatment conditions, correspondingly in using alumina the pH value increased after each treatment condition. ORP changed vise versa i.e., increased with ceramics and decreased with alumina according to treatment serial (one pass, 5 and 10 min), but each value always remained less than control. ST increased with ceramics treatments, but not with alumina. Eventually, EC showed its lower value with the final treatment (10 min fluidization period) in both cases. Azad and Ishikawa (2003) reported the similar findings using different ceramics treatments where pH values increased and ORP decreased in different treatment periods compare with control. But the differentiated values of these parameters were not the same due to different amounts of granular materials and different treatment temperature. Tamura et al. (1999) also showed the differences in different parameters in water activation system using granular ceramics.
For the treatment of tap water with ceramics and alumina irrespective of all conditions, pH increased and ORP decreased compare to control other than ST and EC. The values of pH at control was moderately/slightly acidic (6.96), after treatments with ceramics and alumina it turned moderately alkaline (7.05-7.22) which is more preferable for drinking and agricultural purposes (Ayers and Westcot, 1985). With the same treatment conditions, the tendency of decrement of ORP value from its initial higher (759 mV) level to lower level (becoming lower than 759 mV) also indicated positive signs for drinking and agriculture. Matsuo et al. (1994) examined that the suitable range of ORP for agriculture was 200-300 mV.
Mechanisms: In the fluidization process of water with ceramics and alumina, the state of molecules of water changed (Kubo, 1989). When water flows through these granular substances, every particle comes into contact with each other as well as with the water molecules in a frictional way and electrochemical reactions takes place (Ishikawa and Tamura, 1996). Resultantly, there has been raising the temperature of the disperse medium and oxidation-reduction have been taken place for which increased the level of dissolved oxygen in the medium (Azad and Ishikawa, 2004). These were considered as the possible causes for changing some characteristics of water like pH, ORP, ST and EC in this experiment.
CONCLUSIONS
Ceramics and alumina were used in a fluidization system for treating tap water through the experimental design elements such as volume of fluidization layer, fluidization time, speed of fluidization and the amount of these fluidization substances (ceramics and alumina). Experimental results in using distilled water showed that after each treatment with ceramics and alumina, pH, ST and EC increased while ORP decreased as compared to untreated distilled water in every cases. Alternatively, while using Kochi tap water, pH and ST increased, ORP decreased as compared with untreated tap water and the values of EC did not change significantly in the same experimental conditions. The increasing and decreasing tendencies of pH and ORP, respectively from their initial values in the study were preferable for drinking and agricultural purposes.
REFERENCES
- Azad, A.K. and K. Ishikawa, 2003. Treatments of different water using natural minerals and its influence on early plant growth, Komatsuna (Brassica rapa L. nothovar). Environ. Control Biol., 41: 311-319.
Direct Link - Azad, Md. A.K. and K. Ishikawa, 2004. Effects of fluidized-ceramics treatment on the flocculation of suspension in water treatment system. Food Agric. Environ., 2: 234-237.
Direct Link - Azad, A.K. and K. Ishikawa, 2004. Water treatment system by fluidization electrolysis using granular ceramics. Agric. Med., 134: 216-222.
Direct Link - Bouwer, H., 1994. Irrigation and global water outlook. Agric. Water Manage., 25: 221-231.
Direct Link - Ishikawa, K., M.A.K. Azad and N. Islam, 2005. Role of ceramics treatment in fluidization method to remove scale from hot spring water. J. Biological Sci., 5: 279-282.
CrossRefDirect Link








