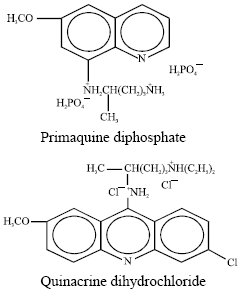
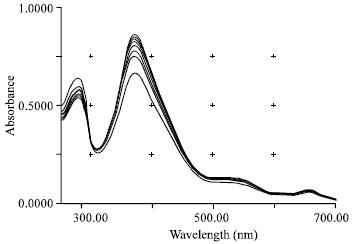
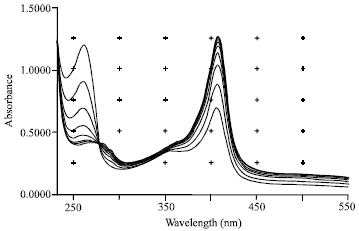
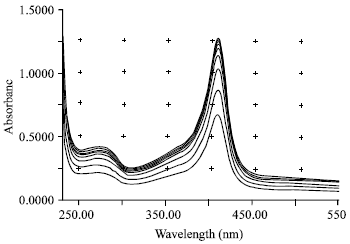
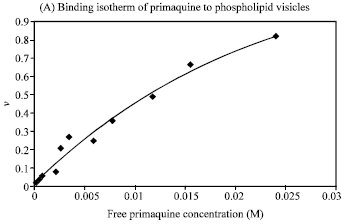
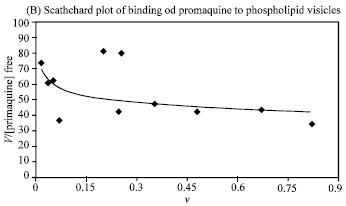
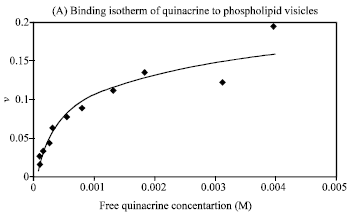
Research Article
Thermodynamic Studies of Antimalarial Drugs and Their Interaction with Myoglobin, Hemoglobin and Phospholipid Model Membranes
Department of Chemistry, College of Arts and Sciences, Arab American University at Jenin, P.O. Box 240, Jenin, Palestine