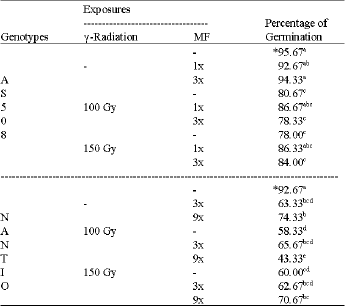
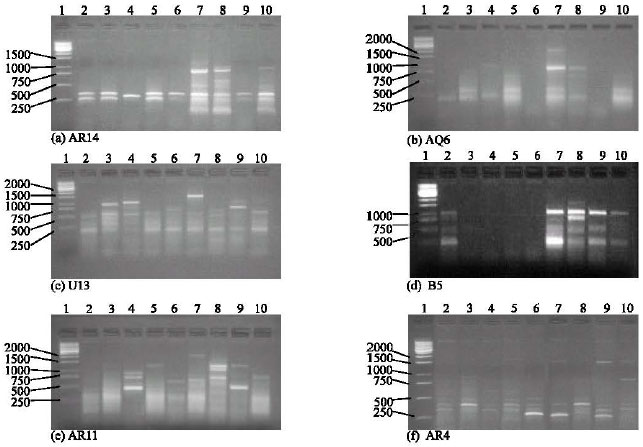
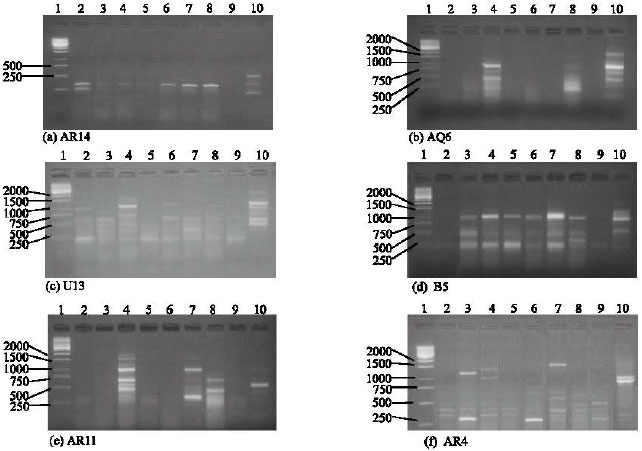
Research Article
Employment of RAPD Technique to Assess the Genetic Stability of Helianthus annuus Treated with Different Mutagenic Agents
College of Health, Onsekiz Mart University, �anakkale, Turkey
Sevil Oldacay
College of Health, Onsekiz Mart University, �anakkale, Turkey