Research Article
Antioxidative Effect of Red Pepper Oil in Edible Oils
Department of Food Sciences and Technology, NWFP Agricultural University, Peshawar, Pakistan
Anwar Ali Shad
Department of Agricultural Chemistry, NWFP Agricultural University, Peshawar, Pakistan
Javid Ullah
Nuclear Institute for Food and Agriculture, Tarnab, Peshawar, NWFP, Pakistan
Taufiq Ahmad
Department of Food Sciences and Technology, NWFP Agricultural University, Peshawar, Pakistan
Qazi Ihsan
Department of Food Sciences and Technology, NWFP Agricultural University, Peshawar, Pakistan









Bibiye, Alaye A. S. Reply
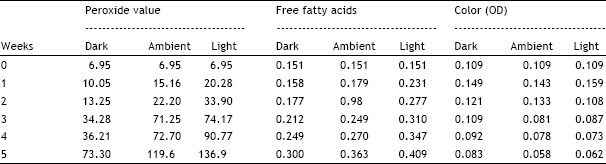
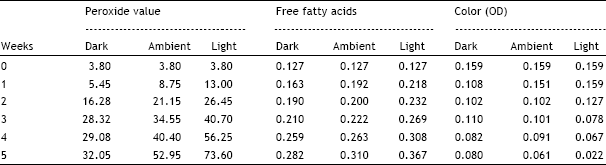
The study has clearly demonstrated the development of rancidity under the influence of fluorescent light as compared to dark light conditions.
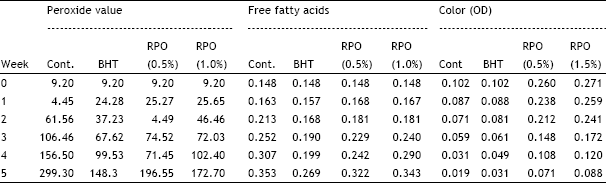
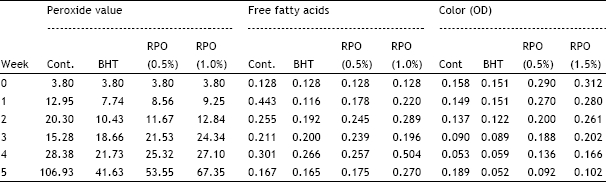
2. The study reveals the efficacy of natural and synthetic anti-oxidants in oils when exposed to light.
3.By comparism, BHT proved a better oxidant than RPO.
However, how this spices (pepper) is relative to other spices was not considered. More so, the source of the edible oils was not define at a glance in the abstract. Above all, bravo for a job well-done.