Research Article
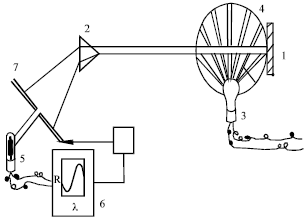
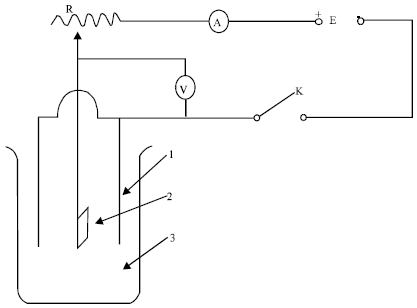
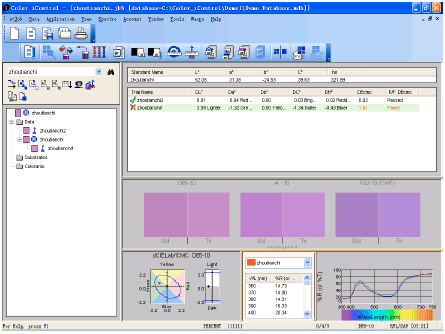
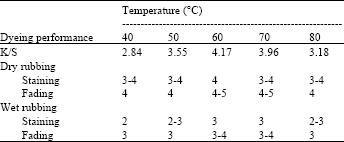
Application Study of Computer Color Measuring Technology Used in the Process of Aluminum Coloring
College of Textile and Garment, Yancheng Institute of Technology, 224051, Yancheng, China
Lu Da-Lian
College of Chemistry, Chemical Engineering and Biotechnology, DongHua University, 201620, Shanghai, China