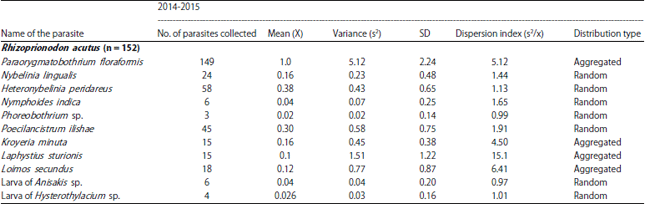
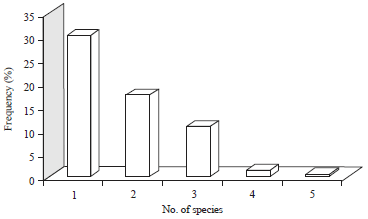
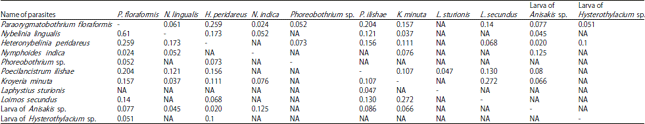
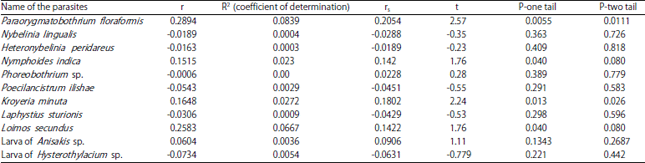
Research Article
Metazoan Parasite Infracommunities of Rhizoprionodon acutus from Nellore Coast off Bay of Bengal Andhra Pradesh, India
Yogi Vemana University, Kadapa, Andhra Pradesh, India
Anu Prasanna Vankara
Yogi Vemana University, Kadapa, Andhra Pradesh, India
LiveDNA: 91.16872