Research Article
Variation in Parasite Infracommunies of Tilapia zillii in Relation to Some Biotic and Abiotic Factors
Department of Zoology, Faculty of Science, Suez Canal University, Ismailia, Egypt
Investigations on parasite communities are useful for knowledge of community structure and dynamics (Esch et al., 1990) and estimating environmental influences (Khan and Thulim, 1991). Parasite community structure may experience changes related to variations in biotic and abiotic factors; these variations can be reflected in species composition and density (Zander, 2005; Tavares and Luque, 2008; Violante-Gonzalez et al., 2010). Parasitic fauna of fishes respond strongly to alterations in the physical and chemical characteristics of the aquatic environment and modifications in the physiological and biological conditions of hosts (Ferrari-Hoeinghaus et al., 2006).
Parasite community structure and species composition may influenced by host-related factors such as diet, body size, reproductive behavior, vagility and migratory habits (Vidal-Martinez and Poulin, 2003; Violante-Gonzalez et al., 2008; Tavares and Luque, 2008). Factors linked to habitat environmental and biological aspects can also affect structure and species composition (Vidal-Martinez and Poulin, 2003; Tavares and Luque, 2008; Violante-Gonzalez et al., 2010).
Ecological studies of fish parasites offer important information not only regarding their hosts but also of the aquatic environment in general through patterns of parasite species occurrences and the prevalence and intensity of parasitism (Ferrari-Hoeinghaus et al., 2006). Various studies have been conducted on parasite communities of fish to analyze parasite communities (Holmes and Price, 1986) with emphasis on how these communities are structured and which processes are involved in maintaining these structures. One of the less studied aspects of fish parasite community ecology is whether or not these communities experience changes in structure and species composition in response to variations in biotic and abiotic factors (Violante-Gonzalez et al., 2008). The objectives of this work were: (1) to compare the parasite community of wild and cultured Tilapia zillii; (2) to compare parasite prevalence and intensity in wild and cultured fish and (3) to study the role of host size (length) and host sex in structuring parasite community.
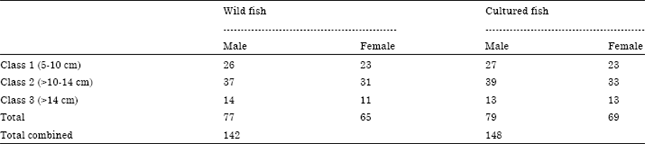
A total of 290 (142 wild fish and 148 cultured fish) Tilapia zillii were collected from lake Manzalah, Egypt (31°15'N; 32°00’E) from July to September 2010. Wild fishes were sampled from Lake Manzalah and cultured ones from farms surrounding Lake Manzalah. These specimens based on their origin were transported in individual holding bags to the laboratory to prevent parasite host-switching and parasite release. The sexes of the fish were determined only after dissecting the fishes and noting the presence of testes or ovaries (Olurin and Somorin, 2006). The data collected for each fish included: (a) host sex; (b) host length classes (class 1: 5-10 cm; class 2: >10-14 cm and class 3: >14 cm) and (c) location of collected fish (wild and cultured fish). The structure of the sampled host population is shown in Table 1. The fish samples were examined for both ectoparasites and endoparasites. For ectoparasites, the external surface of the fish was examined thoroughly. Areas around the fins, nostrils, operculum and the buccal cavity were examined for external parasites (Monogeneans, crustaceans). Gills were removed and examined whole under a dissecting microscope. Gill smears were also made and examined under the microscope. Pieces of gills were placed in 4% formalin in a vial, shaken and the sediment examined under a microscope. Small fish were placed in containers of 4% formalin, shaken and the sediment examined for parasites. For endoparasites, each fish was dissected and the internal organs examined. The entire digestive system was removed and placed in a Petri dish with physiological saline. The gut was divided into sections and each section examined for parasites. The gonads, liver, heart and gall bladder and the pericardial cavity were examined for parasites. If found, each group of parasites was treated as follows: acanthocephalans were placed in distilled water in vials and left overnight in a refrigerator.
| Table 1: | The structure of the sampled host population of wild and cultured Tilapia zillii by host sex and host length classes |
 | |
This caused the proboscis of the acanthocephalans to extrude. Trematodes were fixed in glacial acetic and pressed between two slides. This makes the worms transparent so internal organs can be seen. All parasites were preserved in 70% alcohol after individual treatments (Aloo et al., 2004). Identification of the monogeneans under was carried out according to Ergens (1981) for species of Cichlidogyrus. Gyrodactylus cichlidarum was identified. Crustcean parasites were identified according to the key provided by Bauer (1987). Representative specimens of the parasites collection were deposited in the parasite collection of Zoology department, Faculty of science, Suez Canal University, Egypt.
The parasitological terms follow Bush et al. (1997). Prevalence is the number of infected fish with one or more individuals of a particular parasite species (or taxonomic group) divided by the number of hosts examined (expressed as a percentage); mean abundance (±SE) is the mean number of parasites in all examined fish; intensity is the number of individuals of a particular parasite species in a single infected host and mean intensity is the average intensity or the total number of parasites of a particular species found in a sample divided by the number of infected hosts. Analyses were taken to the component community and infracommunity levels (Holmes and Price, 1986) that is, all the parasite species in all the hosts collected per sampling period and all the parasite species in an individual fish, respectively.
Statistical analysis: Effect of host sex and fish origin on infestation abundance and intensity of each parasite species was examined using Mann-Whitney’s test. The difference in parasite prevalence was tested by using Chi-square tests (χ2). Spearman’s correlation coefficient and the correlation coefficient (r) were used to examine relationships between host length and both intensity and prevalence of infestation for each parasite species. Kruskal-Wallis test was used to test for differences in intensity of infestation among host length classes. The results were considered significant at p<0.05. Berger-Parker Index was used as a measure of numerical dominance. Brillouin Diversity Index (H’) was used as a measure of diversity. All the statistical tests were performed by using the software packages SPSS 17.00 (USA).
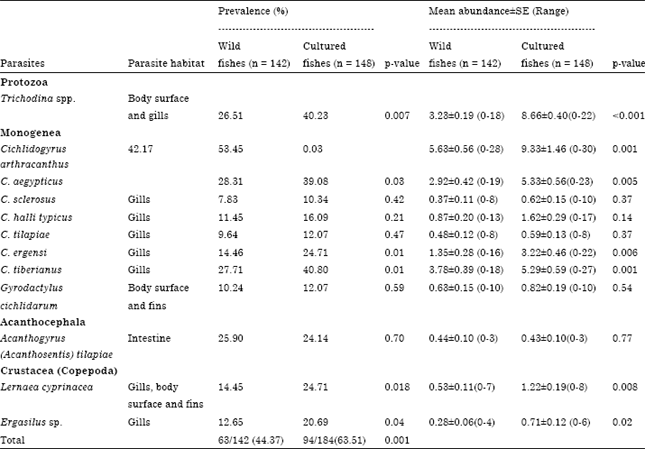
The results showed that, parasite community of wild and cultured, T. zillii consisted of 12 species, one protozoon (Trichodina spp.), eight monogenea (Cichlidogyrus arthracanthus, C. aegypticus, C. sclerosus, C. halli typicus, C. tilapiae, C. ergensi, C. tiberianus and Gyrodactylus cichlidarum), one acanthocephalan (Acanthogyrus (Acanthosentis) tilapiae) and two crustacean species (Lernaea cyprinacea and Ergasilus sp.) (Table 2). The frequency distributions of the parasite species in all the samples showed the community was made up of two groups: common parasites, with prevalence greater or equal to 25% (C. arthracanthus, C. tiberianus, C. aegypticus, Trichodina spp. and A. tilapiae) and rare parasites, with prevalence less than 25% (C. ergensi, L. cyprinacea, Ergasilus sp., C. halli typicus, G. cichlidarum, C. tilapiae and C. sclerosus). The most prevalent parasite was C. arthracanthus (47.94%) and C. aegypticus (33.82%) while the less prevalent one was C. sclerosus (9.12%) in both wild and cultured fishes combined. The most abundant parasite was C. arthracanthus (15.50±0.65) and C. aegypticus (12.29±0.56) while the less abundant one was A. tilapiae (0.43±0.09) in both wild and cultured fishes combined. T. zillii infracommunities are dominated by monogenean species.
| Table 2: | Prevalence and mean Abundance±SE of parasite infracommunities of wild and cultured Tilapia zillii collected from lake Manzalah, Egypt |
 | |
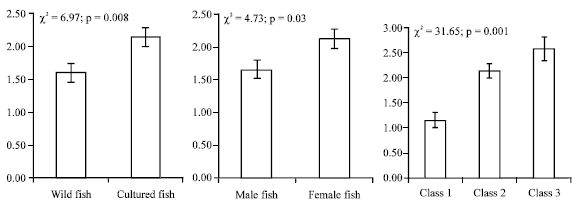 | |
| Fig. 1: | Species richness of parasite infracommunities per origin of fish, host sex and length classes |
The data revealed that host sex, host size (length) and fish origin (wild and cultured) played significant role in determining infracommunity species richness (Fig. 1). The mean species richness of parasite infracommunities harbored per host in cultured fishes was higher (2.13±0.14, χ2 = 6.97; p = 0.008) when compared with that in wild ones (1.87±0.10). The mean species richness was higher in female (2.12±0.15, χ2 = 4.73; p = 0.03) and higher length class fishes (Fig. 1: 2.58±0.23, χ2 = 31.65; p<0.001) when compared with that in male and female, respectively (Fig. 1).
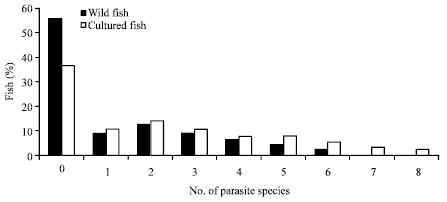 | |
| Fig. 2: | Frequency distribution of infracommunity species richness per host origin (wild and cultured fish) |
The species richness of parasite infracommunities was positively correlated with host body size (R2 = 39; p<0.001). The number of parasite species per infected T. zillii range from 1 to 6 in wild fish and from 1 to 8 in cultured fish. In wild fish, 55.63 had no infection, 9.15% of the hosts were parasitized by a single species while 35.21% had mixed infections: 12.67% had 2 species; 22.54% had from 3-6 and 0% had from 7-8. In cultured fish, 36.48 had no infection, 10.81% of the hosts were parasitized by a single species while 52.70% had mixed infections: 14.18% had 2 species; 32.43% had from 3-6 and 6.08% had from 7-8 (Fig. 2). The diversity (Brillouin index) of parasite community in wild fishes was 2.05 while in cultured fish was 2.08 (p>0.05). The value of Berger-Parker index in wild fish was 0.41 and 0.28 in cultured fish (p = 0.001).
The overall prevalence of infection in cultured T. zillii (63.51%) was significantly higher as compared to that in wild fish (44.37%, χ2 = 7.83; p = 0.001). The prevalence of each species recorded was higher in cultured when compared to that of wild fish except A. tilapiae (Table 2). Significant difference was found in prevalence of infection between wild and cultured fish regarding protozoa (Trichodina spp.), monogenea (Cichlidogyrus arthracanthus, C. aegypticus, C. ergensi and C. tiberianus) and crustacean species (L. cyprinacea and Ergasilus sp.) (Table 2). There were variations in mean abundance of parasites species between wild and cultured T. zillii. The abundance of each species recorded was higher in cultured as compared with that in wild fish except A. tilapiae (Table 2). Significant difference was found in abundance of infection in Trichodina spp. (χ2 = 7.83; p<0.001), C. arthracanthus (χ2 = 12.93; p<0.001), C. aegypticus (χ2 = 8.06; p = 0.005), C. ergensi (χ2 = 7.49; p = 0.006), C. tiberianus (χ2 = 10.06; p = 0.002), L. cyprinacea (χ2 = 6.95; p = 0.008) and Ergasilus sp. (Table 2, (χ2 = 4.98; p = 0.02)).
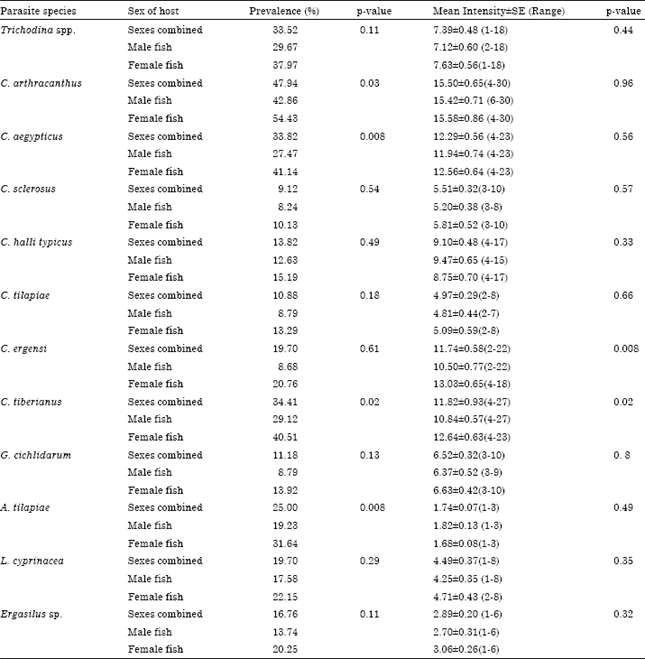
Host sex significantly affects the prevalence of some parasite infracommunities (Table 3). The prevalence of infection was higher in female than that in males in C. arthracanthus (54.43 and 42.86% for female and male fish, respectively), C. aegypticus (41.14 and 27.47% for female and male fish, respectively), C. tiberianus (40.51 and 29.12% for female and male fish, respectively) and A. tilapiae (Table 3; 31.64 and 19.23% for female and male fish, respectively). Moreover, host sex significantly affects the intensity of infection in C. tiberianus. Intensity of infection was higher in female (12.64±0.63) than that in male (Table 3, 10.84±0.57).
| Table 3: | Prevalence and mean Intensity±SE of parasite infracommunities of Tilapia zillii collected from lake Manzalah, Egypt per host sex |
 | |
| Significant level at p<0.05 | |
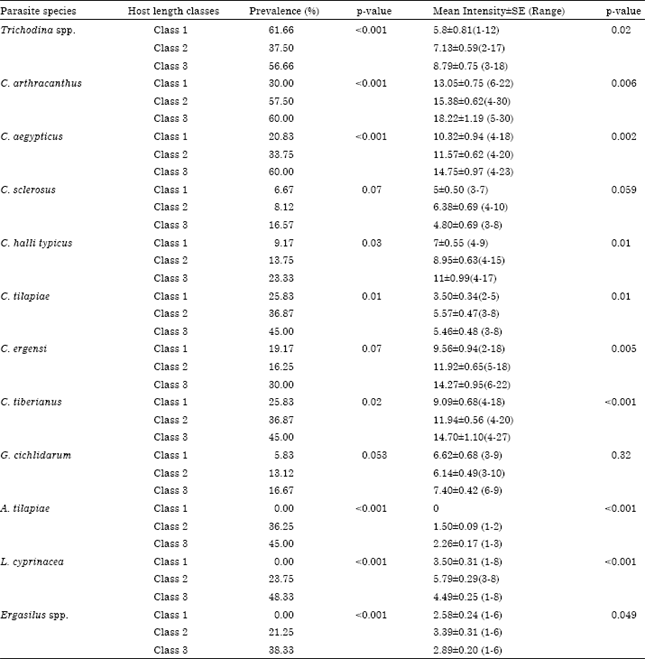
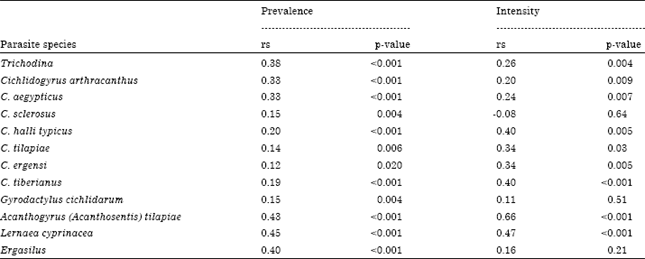
Prevalence of infection was varied significantly by host length class being higher in the length class 3 as follow: Trichodina spp. (56.66%, p<0.001), C. arthracanthus (60%, p<0. 001), C. aegypticus (60%, p<0.001), C. halli typicus (23.33%, p = 0.03), C. tilapiae (45%, p = 0.01), C. tiberianus (45%, p = 0.02), A. tilapiae (45%, p<0.001), L. cyprinacea (48.33%, p<0.001) and Ergasilus sp. (38.33%, p<0.006) (Table 4). Intensity of infection was varied significantly by host length class being higher in the length class 3 as follow: Trichodina spp. (8.79±0.75, p = 0.02), C. arthracanthus (18.22±1.19, p = 0.006), C. aegypticus (14.75±0.97, p = 0.002), C. halli typicus (11±0.99, p = 0.01), C. tilapiae (5.46±0.48, p = 0.01), C. ergensi (14.27, p = 0.005), C. tiberianus (14.70±1.10, p<0.001), A. tilapiae (2.26±0.17, p<0.001), L. cyprinacea (4.49±0.25, p<0.001) and Ergasilus sp. (2.89±0.20, p = 0.048) (Table 4). Based on all samples examined, total host length was positively correlated with prevalence of most species of parasite community of T. zillii (Table 5). Moreover, mean intensity of Trichodina spp., C. arthracanthus, C. aegypticus, C. halli typicus, C. ergensi, C. tilapiae, C. tiberianus, A. tilapiae, L. cyprinacea and Ergasilus sp. were positively correlated with host length of the host (Table 5).
| Table 4: | Prevalence and mean intensity±SE of parasite infracommunities of Tilapia zillii collected from lake Manzalah, Egypt per host length classes |
 | |
| Significant level at p<0.05 | |
| Table 5: | Correlation between host length as continuous factor and both prevalence and intensity of parasite infracommunities of Tilapia zillii |
 | |
| Significant level at p<0.05 | |
DISCUSSION
All species of parasite community that found in this study have been previously reported (Ramadan, 1991; Hagras et al., 2001; El-Naggar and El-Tantawy, 2003). Parasite community of T. zillii shows low diversity and species richness, it can be considered depauperate. In T. zillii, parasite community was generally poor in number of parasite species and lower in diversity compared to other tropical fish species such as Cichlasoma (Nandopsis) urophthalmus (38 species), Parachromis managuensis (34) and Thorichthys hellerin (32) (Salgado-Maldonado et al., 2005; Violante-Gonzalez et al., 2008). Similarly, Kennedy (1990) found that infracommunities of eels were species poor and exhibited low diversity. He suggested that the structure of such infracommunities influenced by stochastic factors: there were empty niche spaces, individual infracommunities were dominated by different species and there was a great deal of variation between them. Each fish appeared to be an independent sampler of the parasites available and evidence for interactions was very weak. Parasite communities in fish can be considered isolationist in nature and to be essentially stochastic assemblages rather than structured communities (Kennedy, 1990). The low species richness in T. zillii may be attributed to physiological, reproductive and feeding behavior factors. Philippart and Ruwet (1982) and Crutchfield et al. (2000) reported that T. zillii primarily feeds on detritus, filamentous algae, aquatic macrophytes and plants of terrestrial origin which may also explain this host’s low intestinal helminth richness (one species). The richest intestinal helminth assemblages in tropical fish are generally observed in hosts with diets based on macroinvertebrates and fish while herbivores and detritivores will generally have poorer intestinal helminth communities (Choudhury and Dick, 2000). T. zillii infracommunities are dominated by monogenean species. The higher prevalence of the monogenea taxa may be due to their direct life cycle. Kennedy et al. (1986) related the poorest parasite community in fish compared to richest communities in birds, to poikilothermy, a poorly differentiated alimentary tract, diet and relatively poor host vagility.
The results indicate that the species richness of parasite infracommunities was positively correlated with host body size. This finding is in consistency with Violante-Gonzalez et al. (2010) and Munoz and Zamora (2011). In general, a larger fish is more likely to be sexually mature and more likely to have been exposed to parasites for a longer time, thereby increasing the probability of larger infrapopulation sizes and greater infracommunity diversity of parasites. Accordingly, larger fish may have more, or different, parasite species than smaller fish (Zapata et al., 2006; Munoz and Zamora, 2011). In addition to, piscine hosts frequently change their behavior and habitat preferences during the aging process. Many fish species shift their diet and habitat requirement at different phases of their lives (Gning et al., 2008). Collectively, these changes can strongly affect the composition, abundance and species richness of parasite community.
The finding that prevalence and mean abundance of parasite were higher in cultured T. zillii fishes than those in wild fishes may be attributed to that, cultured fishes are more concentrated than they are in nature. Obiekezie (1991) noted that in wild fishes, monogeneans occurred at low intensities and in apparent equilibrium with their hosts, whereas under cultured conditions, these pathogens build up heavy worm burdens which provoke epizootics. Similarly, Violante-Gonzalez et al. (2009) found higher infections levels of Diplostomum compactum in cultured tilapia than wild Nile tilapia, O. niloticus. They attributed this to higher fish densities in the culture systems. Monogenea have the highest prevalence and abundance followed by acanthocephalan, protozoa and crustacea. This may be attributed to variation in physico-chemical factors and that fishes are more concentrated in cultured site than these were in wild site. On the other hand, Ayanda (2009) recorded a total prevalence of 27.5% in wild fish samples of Clarias gariepinus and 0% in cultured one. He attributed his results to the improved management practices such as avoidance of overcrowding, poor environmental conditions and pollution and good environmental conditions carried out by fish farmers in the study area.
The prevalence and intensity of infection in most of parasite species were higher in females than those in males of O. niloticus. This observation agrees with the finding of Ramadan (1991) who found that female fishes were generally more parasitized than males in Tilapia nilotica, T. zillii and T. galilaea. Similarly, Ibrahim and Soliman (2011) found that the mean infection intensity of P. ascolonga was higher in females than that in males in T. zillii. The sex differences in infection may be attributed to the immune response of the host due to the difference in endocrine glands activities between the male and female host fishes which have been suggested by Poulin (1996) and Ibrahim and Soliman (2011). The higher prevalence and intensity in female fishes of O. niloticus may be related to investment in reproduction of female fishes is more costly than that in males ones, thus females are more susceptible to parasite infection in periods of investment in gonad development (Simkova et al., 2005). On the contrast, Aloo et al. (2004) reported that males tended to harbor more parasites than females in many freshwater fish species. However, Poulin (1996) concluded that the sex difference is irrelevant and no significant differences between the prevalence and intensity of infection in female and male hosts.
The positive significant correlation of prevalence and mean parasite intensity with total host length indicates that larger hosts harbored more parasite species and more parasite burden than smaller ones. This is attributed to the fact that larger (i.e., older) fish had more time to accumulate parasites than younger (smaller) ones (Bush et al., 2003; Zander, 2004). Boungou et al. (2008) showed no significant difference in the rate of parasitism with monogena according to class of size of O. niloticus. In contrast, Bakke et al. (2002) found a negative correlation between the growths the population of Gyrodactylus salaries and age of salmonidae. They explained their results due to the immune response that increases with the age of fish.
In conclusion, T. zillii aquaculture activities had influence on the relative prevalence and abundance of certain parasite species in addition to species richness of parasite community. Moreover, host traits such as body size and host sex affect parasite community structure. Other studies on T. zillii are needed to provide insight into environment and community level impacts of aquaculture.